Modulating ER morphology and stress to boost recombinant protein accumulation
SUPERVISOR: EVA STÖGER
Background.
Plants are attractive production hosts for biopharmaceuticals, but as with all recombinant systems, yield and quality of product are key determinants for the economic viability of the production process (Stoger et al., 2014). High levels of desired product are not always achieved for a variety of reasons, some reflecting the intrinsic properties of the expressed protein and others reflecting external factors that limit expression and accumulation.
Biological limitations in the plant cell appear particularly relevant for complex proteins requiring optimal conditions for folding and a specific intracellular environment to ensure stability. Most of these proteins pass the endoplasmic reticulum (ER) en route to the apoplast or their intracellular destination. The ER itself can also act as a storage compartment, and retention in the ER is a common strategy to increase the yield of recombinant proteins (Sack et al., 2014; Hofbauer and Stoger, 2013). However, high levels of recombinant protein synthesis place great demands on the host and several reports have described morphological changes of the ER and gene expression patterns indicating an ER stress response (Kudo et al., 2013). Often this also leads to the sequestration of the unfolded proteins into separate compartments resembling the ER-protective organelles (ERPOs), and the plant-produced recombinant proteins can be extracted only partially in a functional, soluble form, whereas a large proportion may be deposited as insoluble aggregates (Vamvaka et al., 2016).
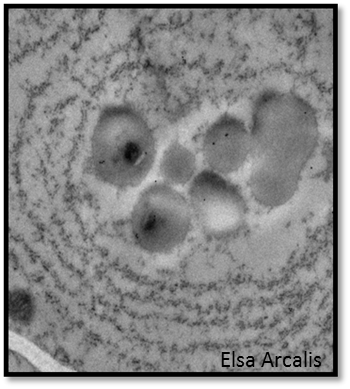
ERPO-like structures were also observed in CHO cells, thus reducing the reported antibody yield (Reinhart et al., 2014). In recombinant yeast and mammalian production platforms, several targets for improving protein folding have been used to reduce ER stress and its damaging effects thus improving the secretion of recombinant proteins (Delic et al., 2014).
While it is not yet known in detail, how the ER structure relates to its function, the ER membrane generally converts from tubules into sheets upon increased secretory load or in differentiating cells (Harris and Chrispeels, 1980). Manipulating the ER architecture along with balancing the redox system and ER stress seem therefore key to increasing its productivity/synthetic capacity as well as its storage capacity.
Aims and methods.
We seek to optimize recombinant product yield and quality through interventions that target the endoplasmic reticulum – the sub-cellular environment for protein production, folding, post-translational modifications and quality control. Morphological changes of the ER are well described during development and due to the expression of ER morphogens such as reticulons (Sparkes et al., 2010, Ibl et al., 2018). Databases will be screened for existing mutants of identified key ER-morphogens, and further Arabidopsis and Nicotiana mutants will be generated by overexpression, CRISPR-Cas-mediated knockout and/or RNAi silencing.
The resulting plants will be analysed by TEM and SBFSEM to reconstruct ER morphology and to determine the abundance of endomembrane-derived organelles. Plants with extensive ER proliferation (as seen for example with knock-out of phosphatidic acid phosphohydrolase PAH1 and 2; Chuang et al., 2014) or with a relative increase of sheet structures will be identified and tested by transient expression of different recombinant target molecules including antibodies and aggregation-prone targets such as interleukin (Morandini et al., 2011). Immunoassays and/or SPR and activity assays will be used for protein quantification.
In addition, the influence of the intracellular redox environment on recombinant protein quality will be monitored and assessed using redox-sensitive GFP constructs, and redox metabolism will be modulated by gene knockout or silencing. Similarly, ER stress markers and transcription factors (e.g. spliced and unspliced versions of bZIP60, BiP family, protein disulfide isomerases 1–1 and 2–3, and calnexin) will be determined upon recombinant protein expression in infiltrated N. benthamiana leaves (in collaboration with STRASSER). Although some aspects of the unfolded protein response (UPR), such as the activation of ERAF (ER-associated folding) pathways, are likely to facilitate the production of functional recombinant proteins, the associated quality control mechanisms and general downregulation of secretory protein expression is likely to have a negative impact on the yield. Accordingly, arm-selective UPR activation will be attempted by generating knock-out mutants of N. benthamiana by CRISPR/Cas-mediated editing of selected key regulators of the UPR. Other transcription factors will be over-expressed by co-infiltration with the construct encoding the recombinant target protein in order to determine a potential effect of pre-emptive and selective induction of ERAF.
Quantitative and qualitative effects on recombinant protein production will be investigated (in collaboration with OBINGER), post-translational modifications such as N-glycosylation of the recombinant proteins will be analyzed (in collaboration with ALTMANN) and their subcellular localization will be determined.
Collaborations within this thesis will include ALTMANN (glycan analysis), OBINGER (spectroscopic and calorimetric techniques) and STRASSER (ER stress markers).
Chuang, C., Barajas, D., Qin, J., Nagy, P.D. (2014) Inactivation of the host lipin gene accelerates RNA virus replication through viral exploitation of the expanded endoplasmic reticulum membrane. PLoS Pathog. 10, e1003944. doi: 10.1371/journal.ppat.1003944
Delic, M., Göngrich, R., Mattanovich, D., Gasser, B. (2014) Engineering of protein folding and secretion-strategies to overcome bottlenecks for efficient production of recombinant proteins. Antioxid. Redox Signal 21, 414-437. doi: 10.1089/ars.2014.5844
Harris, N., Chrispeels, M. (1980) The endoplasmic reticulum of mung-bean cotyledons: Quantitative morphology of cisternal and tubular ER during seedling growth. Planta 148, 293-303. doi: 10.1007/BF00380041
Hofbauer, A. and Stoger, E. (2013) Subcellular Accumulation and Modification of Pharmaceutical Proteins in Different Plant Tissues. Curr. Pharm. Des. 19, 5495-5502. doi: 10.2174/1381612811319310005
Ibl, V., Peters, J., Stoger, E., Arcalís, E. (2018) Imaging the ER and Endomembrane System in Cereal Endosperm. Methods Mol. Biol. 1691, 251-262. doi:10.1007/978-1-4939-7389-7_20
Kudo, K., Ohta, M., Yang, L., Wakasa, Y., Takahashi, S., Takaiwa, F. (2013) ER stress response induced by the production of human IL-7 in rice endosperm cells. Plant Mol. Biol. 81, 461-475. doi: 10.1007/s11103-013-0016-5
Morandini, F., Avesani, L., Bortesi, L., Van Droogenbroeck, B., De Wilde, K., Arcalis, E., Bazzoni, F., Santi, L., Brozzetti, A., Falorni, A., Stoger, E., Depicker, A., Pezzotti, M. (2011) Non-food/feed seeds as biofactories for the high-yield production of recombinant pharmaceuticals. Plant Biotechnol. J. 9, 911-921. doi: 10.1111/j.1467-7652.2011.00605.x
Reinhart, D., Sommeregger, W., Debreczeny, M., Gludovacz, E., Kunert, R. (2014) In search of expression bottlenecks in recombinant CHO cell lines--a case study. Appl. Microbiol. Biotechnol. 98, 5959-5965. doi: 10.1007/s00253-014-5584-z
Sack, M., Hofbauer, A., Fischer, R., Stoger, E. (2014) The increasing value of plant-made proteins. Curr. Opin. Biotech. 32, 163-170. doi: 10.1016/j.copbio.2014.12.008
Sparkes, I. Tolley, N., Aller, I., Svozil, J., Osterrieder, A., Botchway, S., Mueller, C., Frigerio, L., Hawes, C. (2010) Five Arabidopsis reticulon isoforms share endoplasmic reticulum location, topology, and membrane-shaping properties. Plant Cell 22, 1333-1343. doi: 10.1105/tpc.110.074385
Stoger, E., Fischer, R., Moloney, M., Ma, J.K. (2014). Plant Molecular Pharming for the Treatment of Chronic and Infectious Diseases. Annu. Rev. Plant. Biol. 65, 743-768. doi: 10.1146/annurev-arplant-050213-035850
Vamvaka, E., Twyman, R.M., Murad, A.M., Melnik, S., The, A.Y., Arcalis, E., Altmann, F., Stoger, E., Rech, E., Ma, J.K., Christou, P., Capell, T. (2015) Rice endosperm produces an underglycosylated and potent form of the HIV-neutralizing monoclonal antibody 2G12. Plant Biotechnol. J. 14, 97-108. doi: 10.1111/pbi.12360
