SECONDARY CELL WALL POLYMER PYRUVYLATION AS AN ESSENTIAL LIGAND MODIFICATION FOR SLH-DOMAIN RECOGNITION
SUPERVISOR: CHRISTINA SCHÄFFER
Project assigned to: CORDULA STEFANOVIC
Background.
Bacterial cell walls have traditionally been a target of antibiotics. Because of the worldwide spread of antimicrobial resistance and the paucity of novel drugs in the development pipeline, learning about novel concepts of how bacteria stick their cell wall together provides an invaluable source for alternative options for the treatment of infections caused by drug-resistant bacteria.
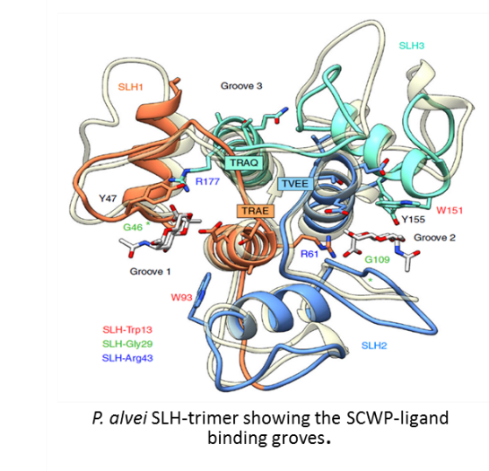 4,6-pyruvateketal-modified β-D-N-acetylmannosamine (Pyr--D-ManNAc) present on several peptidoglycan-bound secondary cell wall polymers (SCWPs) is an essential and sufficient epitope for Gram-positive cell wall anchoring of proteins harboring a terminal S-layer homology (SLH) domain trimer [1]. We recently characterized the structural basis of this anchoring mechanism by analyzing co-crystal structures of the three conse¬cutive SLH domains from the S-layer protein SpaA of the model organism Paenibacillus alvei with synthesized monosaccharide and disaccharide building blocks of its SCWP, which consists of [3)-4,6-Pyr--D-ManpNAc-(14)--D-GlcpNAc-(1] repeats [2]. Notably, a comparable situation is present in the cell wall of pathogenic Bacillus anthracis [3,4]
4,6-pyruvateketal-modified β-D-N-acetylmannosamine (Pyr--D-ManNAc) present on several peptidoglycan-bound secondary cell wall polymers (SCWPs) is an essential and sufficient epitope for Gram-positive cell wall anchoring of proteins harboring a terminal S-layer homology (SLH) domain trimer [1]. We recently characterized the structural basis of this anchoring mechanism by analyzing co-crystal structures of the three conse¬cutive SLH domains from the S-layer protein SpaA of the model organism Paenibacillus alvei with synthesized monosaccharide and disaccharide building blocks of its SCWP, which consists of [3)-4,6-Pyr--D-ManpNAc-(14)--D-GlcpNAc-(1] repeats [2]. Notably, a comparable situation is present in the cell wall of pathogenic Bacillus anthracis [3,4]
Despite the importance of the Pyr-β-D-ManNAc SLH binding interaction for cell wall assembly the necessary pyruvylation reaction of β-D-ManNAc as well as the role of pyruvyl-ation in the transfer reaction of the SCWP to peptidoglycan are widely uncharacterized, both within SCWP biosynthesis and from a biochemical and molecular point of view. This PhD thesis aims at gaining insight into these two key steps of SCWP biosynthesis as a prerequisite for cell wall assembly. Our current perspective of the P. alvei pyruvyltransferase CsaB is the basis for the proposed research: i) CsaB uses PEP as donor substrate and is active on 11-phenoxy¬undecyl-PP-α-D-GlcNAc-(14)--D-ManNAc, but not on synthetic pNP--ManNAc, UDP-ManNAc or -D-ManNAc-(1→4)--D-GlcNAc-O-Me [5]; ii) Ala mutation of amino acid residues predictably involved in PEP binding yielded inactive CsaB; iii) pure CsaB is well behaved in solution and initial crystallization screens with CsaB in complex with PEP yielded several 'hits' of microcrystals (S.V. EVANS). We hypothesize that CsaB-catalyzed -D-ManNAc pyru¬vylation occurs at the stage of the lipid-linked SCWP repeat and has implications on downstream ligation of SCWP to peptidoglycan by an LytR-CpsA-Psr (LCP) enzyme [6-8].
Aims and methods.
By employing a bottom-up approach involving a portfolio of defined synthetic, lipid-linked SCWP precursor fragments (M. BLAUKOPF/P. KOSMA) in concert with recombinant CsaB and a recombinant LCP SCWP-PG ligase the molecular basis of b-D-ManNAc pyruvylation and its status within SCWP biosynthesis shall be unraveled.
Experimental set-up:
As a starting point, recombinant CsaB enzyme and an established pyruvylation assay are available in our lab. Enzymes will be characterized with biophysical methods (CD), binding studies with ligands will be performed (ITC) (C. OBINGER); data will be complemented by enzyme modelling and SCWP-fragment/enzyme docking studies (C. OOSTENBRINK).
Collaborations within this thesis will include: BLAUKOPF/KOSMA (synthesis of SCWP fragments; NMR), OBINGER (protein characterization; CD, ITC), and OOSTENBRINK (SCWP-fragment/enzyme docking studies; modelling). International cooperations include: S.V. EVANS (X-ray crystallography) and E.D. BROWN (B. subtilis LCP mutants).
Collaborating research groups where the PhD student could perform a research stay abroad
Stephen V. EVANS (Department of Biochemistry & Microbiology, University of Victoria, Victoria, BC, Canada; svevans@uvic.ca): X-ray crystallography
Eric D. BROWN (Department of Biochemistry and Biomedical Sciences, McMaster University, Hamilton, ON, Canada; ebrown@mcmaster.ca): B. subtilis LCP mutants and assay
1. Hager, F.F., Sützl, L., Stefanović, C., Blaukopf, M., Schäffer, C. (2019) Pyruvate substitutions on glycoconjugates. Int. J. Mol. Sci. 20, pii: E4929. doi:10.3390/ijms20194929
2. Blackler, R.J., López-Guzmán, A., Hager, F.F., Janesch, B., Martinz, G., Gagnon, S.M.L., Haji-Ghassemi, O., Kosma, P., Messner, P., Schäffer, C., Evans, S.V. (2018) Structural basis of cell wall anchoring by SLH domains in Paenibacillus alvei. Nat. Commun. 9, 3120. doi:10.1038/s41467-018-05471-3
3. Chapman, R.N., Liu, L., Boons, G.J. (2018) 4,6-O-pyruvyl ketal modified N-acetyl mannosamine of the secondary cell polysaccharide of Bacillus anthracis is the anchoring residue for its surface layer proteins. J. Am. Chem. Soc. 140, 17079−17085. doi:10.1021/jacs.8b08857
4. Sychantha, D., Chapman, R.N., Bamford, N.C., Boons, G.J., Howell, P.L., Clarke, A.J. (2018) Molecular basis for the attachment of S-layer proteins to the cell wall of Bacillus anthracis. Biochemistry 57, 1949-1953. doi:10.1021/acs.biochem.8b00060
5. Hager, F.F., Lopez-Guzman, A., Krauter, S., Blaukopf, M., Polter, M., Brockhausen, I., Kosma, P., Schäffer, C. (2018) Functional characterization of enzymatic steps involved in pyruvylation of bacterial secondary cell wall polymer fragments. Front. Microbiol. 9, 1356. doi:10.3389/fmicb.2018.01356
6. Schaefer, K., Matano, L.M., Qiao, Y., Kahne, D., Walker, S. (2017) In vitro reconstitution demonstrates the cell wall ligase activity of LCP proteins. Nat. Chem. Biol. 13, 396-401. doi:10.1038/nchembio.2302
7. Gale, R.T., Sewell, E.W., Garrett, T.A., Brown, E.D. (2014) Reconstituting poly(glycerol phosphate) wall teichoic acid biosynthesis in vitro using authentic substrates. Chem Sci 5, 3823-3830. doi:10.1039/C4SC00802B
8. Liziewski Zilla, M., Chan, Y.G.Y., Lunderberg, J.M., Schneewind, O., Missiakas, D. (2015) LytR-CpsA-Psr enzymes as determinants of Bacillus anthracis secondary cell wall polysaccharide assembly. J. Bacteriol. 197, 343-353. doi:10.1128/Jb.02364-14
9. Higuchi, Y., Yoshinaga, S., Yoritsune, K., Tateno, H., Hirabayashi, J., Nakakita, S., Kanekiyo, M., Kakuta, Y., Takegawa, K. (2016) A rationally engineered yeast pyruvyltransferase Pvg1p introduces sialylation-like properties in neo-human-type complex oligosaccharide. Sci. Rep. 6, 26349. doi:10.1038/srep26349
