Instructional Courses
BioToP offers a range of eleven different INSTRUCTIONAL COURSES on special topics within the field of production, purification and structure-function analysis of recombinant proteins produced in various expression systems. Students can choose two courses related to the topic of their PhD thesis and will get hands-on-training on methods and instruments necessary for their work.
IC IA - SPECTROSCOPIC AND CALORIMETRIC ANALYSIS OF PROTEINS
Practical application of circular dichroism and fluorescence spectroscopy as well as differential scanning calorimetry in analysis of protein (un)folding pathway(s), structure, conformational and thermal stability and mutant screening
Outcomes: Knowledge of strengths and shortcomings of various spectroscopic and calorimetric techniques in elucidation of protein purity, stability and structure, protein misfolding, ligand binding and screening techniques.
IC IB - PROTEIN DESIGN BY DIRECTED EVOLUTION
Protein modelling and mutagenesis strategies, design and introduction of selected mutations by saturation mutagenesis, transformation and expression in and purification from E. coli, development of screening assays, multiwell plate-based screening and selection, structural and functional characterisation of selected variants.
Outcomes: Participants should be able to select appropriate directed evolution methodology, to design and apply multiwell plate-based assays and adapt these assays to various proteins, and to use structural and kinetic data for the comparison of enzymes.
IC IIA - MASS SPECTROMETIC ANALYSIS OF PROTEINS AND PROTEOMICS
Theory and practical application of state-of the art methods of protein analysis with emphasis on mass spectrometry. Separation and analysis of proteins (2D-PAGE), ionisation of biomolecules, types of mass analyzers, tandem mass spectrometry, modern MS concepts, identification of proteins by MS and MS-MS, other applications of mass spectrometry; analysis of protein-linked oligosaccharides by HPLC and mass spectrometry. Proteomics and glycoproteomics applications.
Outcomes: Ability to select the optimal analytical methods for a given protein. Knowledge of the potentials and challenges of proteome studies.
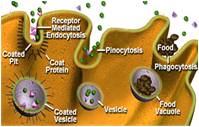
IC IIB - PROTEIN SECRETION AND ENDOCYTOSIS
Expression of a recombinant protein in animal cells, characterization of its subcellular localization, determination of its secretion kinetics and half-life, assessment of its fate after endocytosis. Methods to be applied include cell culture and transfection, functional assays, metabolic labeling, immunoprecipitation, electrophoresis, immunoblotting, density gradient centrifugation, immunofluorescence microscopy.
Outcomes: Ability to assess the secretion, uptake and turnover of recombinant proteins produced in eukaryotic cells. Insights into the complex compartmentalization of mammalian cells.
IC IIC - CELL IMAGING
Use of fluorescent protein tags as reporters for subcellular localization, expression of fluorescently-tagged marker proteins in different subcellular compartments in plants and mammalian cells. Methods to be applied include live-cell imaging, confocal laser scanning microscopy, quantitative co-localization studies, measurement of protein dynamics using fluorescence recovery after photobleaching or photoactivatable fluorescent proteins, monitoring of in vivo protein-protein interactions using fluorescent protein techniques like biomolecular fluorescence complementation.
Outcomes: Students will be familiar with basic and cutting edge light microscopy techniques. Increased understanding of the subcellular organization and secretory pathway ofeukaryotic cells.
IC IIIA - ANIMAL CELL CULTURE AND TECHNOLOGY
Establishment of cell cultures of various tissues and species, analysis of viability and apoptosis, stress resistance, analysis of cell characteristics including surface marker profiles by flow cytometry and fluorescence microscopy, cell sorting for subcultivation.
Outcomes:Students will be made familiar with various animal cell cultures and associated advanced technologies. Insights into the individual potential of different animal cell systems for the production of recombinant proteins.
IC IIIB - APPLIED VIROLOGY
Description of various viral replicational strategies as well as host-virus interactions; theory and practical applications of viral systems for protein expression, gene delivery and vaccine production; described examples will include various insect, plant and mammalian viruses.
Outcomes: Understanding of viral strategies and their principal life cycles; ability to choose a suitable viral system for protein expression and gene delivery applications; understanding the rationale behind vaccine design.
IC IVA - MOLECULAR MODELLING
An overview on the principles of biophysical description of proteins in the context of biochemical engineering will be given using defined proteins/enzymes investigated in BioToP projects: structure and stability engineering and modelling; demonstration of complementarity between experimental (biospectroscopy, calorimetry) and computational approaches; concepts of molecular simulation and inhibitor design; useful computer programmes.
Outcomes: Students will be trained in the principles of analysis and prediction of protein structure, prediction of the effects of protein engineering on protein structure, conformational stability and function. Knowledge of state-of-the-art concepts in molecular simulation of protein-ligand interactions and in silico design of inhibitors. Hands-on training in relevant computer programmes.
IC IVB - NEXT-GENERATION SEQUENCING
This course will introduce the students to next-generation sequencing (NGS) technologies, combining theoretical sessions illustrating sequencing concepts, principal library preparation methods, and data analysis strategies. Practical work will start with quality control (QC) and quantitation of genomic DNA, followed by preparation and QC of a genomic library for sequencing on the Illumina NGS platform. Bioinformatics work will include an introduction to read filtering, read mapping, and the analysis of genomic and transcriptome sequencing data.
Outcomes: The course will provide students with an introduction and an overview to the rapidly developing field of NGS. The students will be trained to realize the perspectives how NGS data can impact their research. Based on the knowledge they acquire, the students will be able to make decisions which sequencing strategy to choose for particular research questions, and how to avoid and solve problems frequently encountered during NGS data analysis.
IC IVC - PROTEIN PURIFICATION
An introduction to the governing principles of preparative chromatography, membrane filtration and precipitation/flocculation with respect to separation of proteins and other macromolecules will be given. Relevant methods will be described for the purification of proteins from different starting materials such as microbial cells, mammalian cells and plant tissue, in particular when a purification tag is not available. Also a detailed insight into available purification tags will be provided, and their advantages and disadvantages will be discussed.
Outcomes: The course will provide instructions on how to optimize a purification scheme, set-up a de novo purification protocol without prior knowledge of protein properties and how to scale up a protein purification method. The students will be trained in trouble shooting of chromatography, membrane filtration, precipitation/flocculation and strategies to avoid protein degradation during purification. Students will be able to select purification tags on a rational basis
IC IVD - Applied Statistics for Life Sciences
This course will refresh the principles of statistical data analysis. Topics covered will include: Scales for variables (categorical, numerical), corresponding distributions, descriptive statistics; Visualization of multivariate data sets; Statistical inference – confidence intervals, parametric & non-parametric tests in the one-, two- and many-sample situations; Simple and multiple linear regression and their extensions (categorical predictors, polynomial regression, interaction terms), model diagnostics, variable selection; Logistic regression for binary target variables.
Outcomes: Participants of the course will recognize the need for a statistical treatment or analysis of their data and learn to translate research questions into statistical hypotheses, to choose the correct method(s) and to perform these steps using the statistical software environment R.