Molecular engineering of baculoviral based protein expression in insect cells
SUPERVISOR: REINGARD GRABHERR
Project assigned to: KRISZTINA KOCZKA
Background.
The baculovirus insect cell expression system is an integral component of recombinant protein expression technologies. A variety of complex eukaryotic proteins have been successfully expressed in insect cells Sf9 at large scale including commercialized vaccines. Recently, Trichoplusia ni cell lines (Hi5, TnAO38) have been recognized as an alternative host for their superior capacity in secreting heterologous proteins at high yields. These cell lines have been shown to be feasible to express complex glycosylated proteins, e.g. viral surface proteins and cellular membrane proteins as well as multi-subunit complexes. Insect cell expression systems offer unique advantages over microbial expression system, as they provide protein folding and posttranslational modifications more akin to mammalian cells as compared to microbial cells. In particular, the successful expression of multi-subunit vaccines such as virus like particles has proven the potential of this system (Krammer and Grabherr, 2010). New emerging production cell lines are derived from Trichoplusia ni, as they have been proven to be highly feasible for serum free cell culture and superior for high yield production of secreted glycoproteins. To date, for the generation of many products, also at large scale, mostly Sf9 cells are being used. Thus, little is known about process engineering of up-scaled insect cell based systems based on Trichoplusia ni derived cell lines. Further bottlenecks are reduced yield by stress response to viral infection and proper purification schemes that guarantee consistent cell and baculovirus free products, especially in the case of secreted virus like particles. To date, genomic sequence data are limited for Spodoptera frugiperda and no genomics or transcriptomics data are yet available for Trichoplusia ni.
Aims and methods.
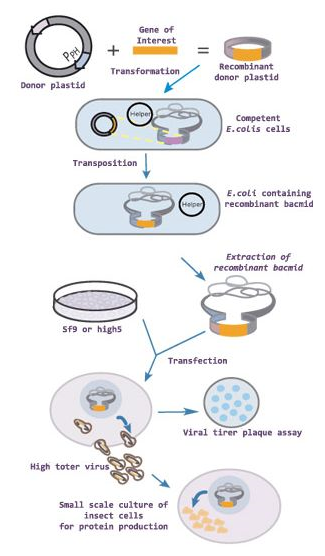
Heterologous protein expression in insect cells is based on infections with transgenic baculoviruses. Depending on the target protein, product yields can vary substantially. Unlike in non-viral protein expression systems, where the cellular machinery is being demanded by the foreign gene expression cassette only, these insect cells are additionally challenged by a massive viral infection causing cell death after 4-7 days. One of the main burdens during the infectious cycle is the unfolded protein response (UPR), assumingly decreasing product yields drastically. Our hypothesis suggests that by modulating key players in e.g. the UPR, production can be prolonged, protein expression improved and the over-all yield markedly increased. To prove this, we will apply two different approaches. First, certain chaperones, folding enzymes, transcriptional factors and pro-survival molecules will be overexpressed, that possibly have an increasing effect on the heterologous protein production, based on the literature. Second, expression of cDNA libraries will serve to over-express/down-regulate each single gene of the overall transcriptome. After identification of regulatory factors that have impact on either yield or quality of a recombinant model protein, these will serve to engineer baculoviruses that together with the target gene express appropriate additional factors. Alternatively, stable engineered T. ni cells will be generated, containing and expressing the same genes. Both strategies will be tested in production processes and downstream processing.
Krammer, F., Grabherr, R. Alternative influenza vaccines made by insect cells. Trends in molecular medicine 16, 313-320 (2010).
