Extracellular vesicle trafficking in aging
SUPERVISOR: JOHANNES GRILLARI
Background.
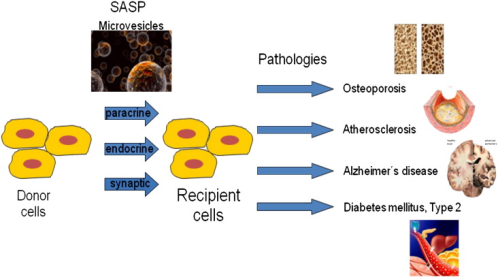
Extracellular vesicles (EVs) are secreted by various cells and have recently raised enormous interest as they carry protein, mRNA and miRNA in an endocrine or paracrine way (Mathivanan et al., 2010), as they can cross the blood brain barrier and thus can be used as therapeutic carrier (Alvarez-Erviti et al., 2011; EL Andaloussi et al., 2013), and as they seem to be the active fraction of mesenchymal stem cell based therapies for e.g. cardiovascular disease (Lee et al., 2013). However, they can also confer negative effects to recipient cells (Schraml et al. in revision; Weilner et al., 2013).
EVs seem to act as a natural ‘lipofection’ agent as they transport active mRNA and miRNAs from a secreting cell to a recipient cell. Interestingly, not much is known about the uptake of EVs into recipient cells, as so far only few proteins mediating uptake have been identified e.g. CD81(Hazawa et al., 2014) and syncytin (Vargas et al., 2014). We recently have observed that EVs from endothelial cells are not taken up by ECs but by MSCs in vitro. This uptake was dependent on endocytosis, since overexpression of a dominant negative form of dynamin inhibited vesicle uptake into MSCs. Therefore, we here propose that uptake might be dependent on receptor-mediated endocytosis and propose to identify ligands on EVs mediating uptake.
Aims and methods.
Since EVs carry cargo that in some instances negatively influences the behaviour of recipient cells, we here will identify neutralizing antibodies to microvesicles secreted by senescent endothelial cells that we have found to inhibit osteogenesis (Schraml et al., in revision, and patent EP10163604.1). To this end, we will establish EV purification strategies in cooperation with JUNGBAUER, establish cell lines that secrete recombinant EVs carrying genetically fused surface proteins, like the EV marker proteins CD81 and CD63, to traceable and affinity-purifiable tags like gfp or Click-tag carrying also protease cleavage sites for mild elution. Native and recombinant EVs bound to beads will be used as antigen to produce single chain antibodies using the Sheet’s single chain Fv (scFv) library (Sheets et al., 1998) in cooperation with RÜKER and OBINGER. As a control, antibodies against CD81 will be established and commercially available ones will be tested for neutralizing activity. These antibodies will then be tested for their ability to inhibit EV uptake in recipient cells (MSCs)in vitro. The antibodies neutralizing EVs will then be used to identify the ligand of EVs that mediate binding to MSCs. The ligand in turn can then be used to identify the receptor on the surface of MSCs.
Therefore, at the end of this project, we will have identified mechanisms of EV uptake by recipient cells, will have established purification and visualisation strategies for EVs, and finally will have identified inhibitors of EV uptake, which per se might be used as therapeutics in cases where vesicle uptake by recipient cells leads to negative effects as in the case of senescent endothelial cell derived microvesicles that inhibit osteogenesis, a mechanism that might contribute to osteoporosis and slow bone healing of the elderly.
Alvarez-Erviti, L., Seow, Y., Yin, H., Betts, C., Lakhal, S., Wood, M.J.A. (2011) Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat. Biotechnol. 29, 341–345
EL Andaloussi, S., Lakhal, S., Moeger, I., Wood, M.J.A. (2013) Exosomes for targeted siRNA delivery across biological barriers. Adv. Drug Deliv. Rev. 65, 391-397
Hazawa, M., Tomiyama, K., Saotome-Nakamura, A., Obara, C., Yasuda, T, Gotoh, T., Tanaka, I., Yakumaru, H., Ishihara, H., Tajima, K. (2014) Radiation increases the cellular uptake of exosomes through CD29/CD81 complex formation. Biochem. Biophys. Res. Commun. 446, 1165–1171
Lee, J.-K., Park, S.-R., Jung, B.-K., Jeon, Y.-K., Lee, Y.-S., Kim, M.-K., Kim, Y.-G., Jang, J.-Y., Kim, C.-W. (2013) Exosomes derived from mesenchymal stem cells suppress angiogenesis by down-regulating VEGF expression in breast cancer cells. PLoS One 8:e84256
Mathivanan, S., Ji, H., Simpson, R.J. (2010) Exosomes: extracellular organelles important in intercellular communication. J. Proteomics 73, 1907–1920
Sheets, M.D., Amersdorfer, P., Finnern, R., Sargent, P., Lindquist, E., Schier, R., Hemingsen, G., Wong, C., Gerhart, J.C., Marks, J.D., Lindqvist, E. (1998) Efficient construction of a large nonimmune phage antibody library: the production of high-affinity human single-chain antibodies to protein antigens. Proc. Natl. Acad. Sci. USA 95, 6157–6162
Vargas, A., Zhou, S., Ethier-Chiasson, M., Flipo, D., Lafond, J., Gilbert, C., Barbeau, B. (2014) Syncytin proteins incorporated in placenta exosomes are important for cell uptake and show variation in abundance in serum exosomes from patients with preeclampsia. FASEB J., in press
Weilner, S., Schraml, E., Redl, H., Grillari-Voglauer, R., Grillari, J. (2013) Secretion of microvesicular miRNAs in cellular and organismal aging. Exp. Gerontol. 48, 626–633
