Recreation of ancestral members of the GMC family of FAD-dependent oxidoreductases
PRINCIPAL INVESTIGATOR: DIETMAR HALTRICH
Background.
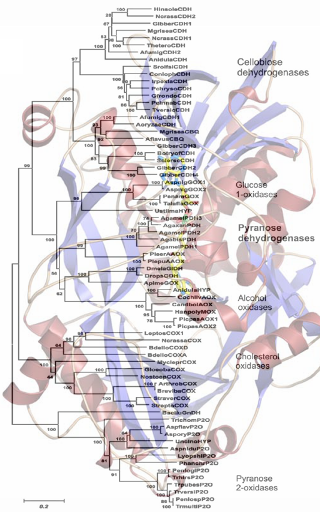 Flavin-dependent enzymes have been implicated in a number of biological processes. They play a central role in aerobic metabolism through their ability to catalyze both one- and two-electron transfer reactions, and take part in various oxidation/reduction reactions in the energy metabolism, but also processes related to DNA repair, protein folding or light emission. Despite this broad spectrum of activities, flavin-dependent enzymes can be grouped into relatively few classes based on their structural and biochemical properties. One of these is the family of glucose-methanol-choline (GMC) oxidoreductases. It shares an evolutionary conserved ca. 30 amino acid sequence comprising a β-α-β motif that plays a role in nucleotide binding as well as five additional blocks of conserved sequences dispersed throughout the primary sequence, which supports the hypothesis of a common evolutionary origin for members of this family (Cavener, 1992). Members of the GMC family perform regiospecific oxidations of the CH-OH moiety of various substrates, and employ oxygen and/or alternative compounds (quinones, dyes, complexed metal ions) as their electron acceptors. Members of the GMC family, for which mechanistic and structural information is available, include glucose oxidase, glucose dehydrogenase, pyranose oxidase, pyranose dehydrogenase, cellobiose dehydrogenase, aryl-alcohol oxidase, and cholesterol oxidase. Some of these enzymes are phylogenetically close to each other, catalyse very similar reactions, yet some (e.g. glucose oxidase, pyranose oxidase) utilise oxygen very efficiently while others (glucose dehydrogenase, pyranose dehydrogenase, cellobiose dehydrogenase) show only very poor reactivity with oxygen (Zamocky et al., 2004). Recently, our group studied the modulation of oxygen reactivity of various GMC members by semi-rational approaches, targeting amino acid residues in the close vicinity of the isoalloxazine ring by saturation mutagenesis (Sygmund et al., 2013; Krondorfer et al. 2014, Brugger et al., 2014). While the Trametes multicolor pyranose oxidase variants T166R, Q448H, L545C, L547R and N593C showed decreased to negligible oxidase and maintained dehydrogenase activity (thus were transformed from an oxidase to a dehydrogenase), we could only identify one mutation each that introduced oxygen reactivity, albeit only to a modest degree, into pyranose dehydrogenase as well as cellobiose dehydrogenase. This indicates that oxygen reactivity is a complex reaction, and cannot be easily introduced into an enzyme by using protein engineering.
Flavin-dependent enzymes have been implicated in a number of biological processes. They play a central role in aerobic metabolism through their ability to catalyze both one- and two-electron transfer reactions, and take part in various oxidation/reduction reactions in the energy metabolism, but also processes related to DNA repair, protein folding or light emission. Despite this broad spectrum of activities, flavin-dependent enzymes can be grouped into relatively few classes based on their structural and biochemical properties. One of these is the family of glucose-methanol-choline (GMC) oxidoreductases. It shares an evolutionary conserved ca. 30 amino acid sequence comprising a β-α-β motif that plays a role in nucleotide binding as well as five additional blocks of conserved sequences dispersed throughout the primary sequence, which supports the hypothesis of a common evolutionary origin for members of this family (Cavener, 1992). Members of the GMC family perform regiospecific oxidations of the CH-OH moiety of various substrates, and employ oxygen and/or alternative compounds (quinones, dyes, complexed metal ions) as their electron acceptors. Members of the GMC family, for which mechanistic and structural information is available, include glucose oxidase, glucose dehydrogenase, pyranose oxidase, pyranose dehydrogenase, cellobiose dehydrogenase, aryl-alcohol oxidase, and cholesterol oxidase. Some of these enzymes are phylogenetically close to each other, catalyse very similar reactions, yet some (e.g. glucose oxidase, pyranose oxidase) utilise oxygen very efficiently while others (glucose dehydrogenase, pyranose dehydrogenase, cellobiose dehydrogenase) show only very poor reactivity with oxygen (Zamocky et al., 2004). Recently, our group studied the modulation of oxygen reactivity of various GMC members by semi-rational approaches, targeting amino acid residues in the close vicinity of the isoalloxazine ring by saturation mutagenesis (Sygmund et al., 2013; Krondorfer et al. 2014, Brugger et al., 2014). While the Trametes multicolor pyranose oxidase variants T166R, Q448H, L545C, L547R and N593C showed decreased to negligible oxidase and maintained dehydrogenase activity (thus were transformed from an oxidase to a dehydrogenase), we could only identify one mutation each that introduced oxygen reactivity, albeit only to a modest degree, into pyranose dehydrogenase as well as cellobiose dehydrogenase. This indicates that oxygen reactivity is a complex reaction, and cannot be easily introduced into an enzyme by using protein engineering.
Aims and methods.
Flavins are among a limited number of organic and inorganic biocatalysts that can effectively reduce oxygen (O2), a hydrophobic molecule by nature, to superoxide (O2−•), hydrogen peroxide (H2O2), or water (H2O). In solution the reaction of reduced flavin with O2 is autocatalytic but also very slow, primarily because the initial single electron transfer from the singlet flavin hydroquinone to the triplet O2 is catalyzed poorly. In flavin-dependent enzymes, O2 reduction can be accelerated by 3–4 orders of magnitude or, alternatively, be abated, indicating that the protein microenvironment controls and modulates flavin reactivity (Massey, 1994; Chaiyen et al., 2012). It was suggested that amongst others a positive charge in the vicinity of the isoalloxazine, a hydrophobic site close to the flavin C(4a) as well as accessibility of the active site through tunnels and channels may be important aspects for oxygen reactivity of flavin-dependent enzymes, yet no general pattern can be given as to the positive or negative modulation of this reactivity with oxygen. Glucose or sugar-oxidizing enzymes of the GMC family apparently developed from a common ancestor. This ancestor could have had both moderate oxidase and dehydrogenase activity; alternatively, improved oxygen reactivity could have been introduced only during evolution of this ancestor towards true oxidases.
It is the aim of this project to reconstruct ancestral genes of these GMC members and to use these as the appropriate background in which to study the effects of historical mutations on functional diversification in oxidases and dehydrogenases, hence to better understand oxygen reactivity / modulation in flavin-dependent enzymes. Phylogenetic methods based on maximum likelihood analysis will be used to construct phylogenetic trees of GMC members, and to infer ancestral sequences at different nodes that connect the paralogous families, mainly those that react with glucose (glucose oxidase GOx, glucose dehydrogenase GDH, pyranose oxidase POx, pyranose dehydrogenase PDH, cellobiose dehydrogenase CDH) (Thornton, 2004). Here nodes between closely related members of the GMC family (GOx/GDH, GOx/PDH, POx/PDH) will be of special interest. These predicted ancestors will then be expressed in a suitable organism (E. coli, P. pastoris) using synthetic genes and biochemically studied in detail. In addition, the target enzyme PDH will be diversified by combinatorially incorporating back-to-ancestor mutations (PETERBAUER) at positions near the alloxazine ring (Goldsmith and Tawfik, 2013). Thereby we aim to understand which mutations will contribute to switching the true dehydrogenase PDH to an oxidase.
Obtained variants will be characterized biochemically and kinetically (OBINGER, CHAIYEN), with the proposed research work including (i) elucidation of the spectral properties of GMC ancestors and PDH variants by using various spectrometric methods (UV-Vis, fluorescence, CD spectroscopy, EPR); (ii) steady-state kinetic analysis; (iii) rapid kinetic studies to see the effect of certain mutations on both the reductive and oxidative half-reaction; (iv) redox potential determination of the obtained proteins / variants using an optically transparent thin-layer electrode cell; (v) measurement of thermodynamic and kinetic stability of wt and variant PDH (determination of melting temperature and half-life of activity); (vi) determination of the crystal structure (wt and variants) in collaboration with the structural biology laboratory at KTH (DIVNE).
Brugger, D., Krondorfer, I., Shelswell, C., Huber-Dittes, B., Peterbauer, C.K., Haltrich, D. (2014) Engineering pyranose 2-oxidase for modified oxygen reactivity. PLoS ONE 9, e109242
Cavener, D.R. (1992) GMC oxidoreductases. A newly defined family of homologous proteins with diverse catalytic activities. J. Mol. Biol. 223, 811–814
Chaiyen, P., Fraije, M.W., Mattevi, A. (2012) The enigmatic reaction of flavins with oxygen. Trends Biochem. Sci. 37, 373–380
Goldsmith, M, Tawfik, D.S. (2013) Enzyme engineering by targeted libraries. Meth. Enzymol. 523, 257–283
Krondorfer, I., Lipp, K., Brugger, D., Staudigl, P., Sygmund, C., Haltrich, D., Peterbauer, C.K. (2014) Engineering of pyranose dehydrogenase for increased oxygen reactivity. PLoS One 9:e91145
Massey, V. (1994) Activation of molecular oxygen by flavins and flavoprotein. J. Biol. Chem. 269, 22459–22462
Sygmund, C., Santner, P., Krondorfer, I., Peterbauer, C.K., Alcalde, M., Nyanhongo, G.S., Guebitz, G.M., Ludwig, R. (2013) Semi-rational engineering of cellobiose dehydrogenase for improved hydrogen peroxide production. Microb. Cell Fact. 12, 38
Thornton, J.W. (2004) Resurrecting ancient genes: experimental analysis of extinct molecules. Nat. Rev. Genet. 5, 366-375
Zamocky, M., Hallberg, M, Ludwig, R., Divne, C., Haltrich, D. (2004) Ancestral gene fusion in cellobiose dehydrogenases reflects a specific evolution of GMC oxidoreductases in fungi. Gene 338, 1–14
