Analysis of structure−function relationships of monomeric and dimeric human IgA1 and IgA2 with defined N− and O−glycosylation
JOINT-SUPERVISORS: Richard STRASSER, Christian OBINGER, Rupert TSCHELIESSNIG
Background.
Immunoglobulin A (IgA) − the most prevalent antibody class at mucosal sites in the human body − is increasingly gaining attention as biopharmaceutical for treatment of infections and cancer (Boross et al., 2013). However, the full potential of recombinant IgAs as therapeutic antibodies is still not fully explored, owing to the fact that (i) robust recombinant production is challenging and (ii) a detailed understanding of structure−function relationships in the two human IgA subclasses, termed IgA1 and IgA2, and their molecular formats [monomeric, dimeric (dIgA1, dIgA2) or secretory forms (sIgA1, sIgA2)] is still lacking. IgA is the most hydrophilic of the Igs reflecting its relatively high level of glycosylation. These distinct structural properties lend this antibody class a unique array of functional capabilities.
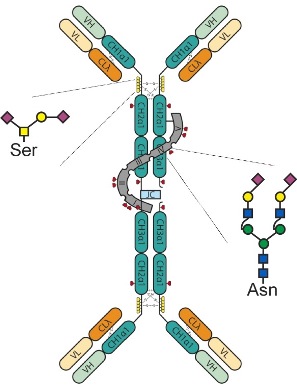
IgAs are extensively glycosylated. Despite the recognized importance of glycosylation for IgG function (Jefferis, 2009) comparatively little is known about the role of glycosylation for the structure of the various IgA formats as well as for their biophysical and immunological properties. The two IgA isotypes (IgA1 and IgA2) carry two to five N−glycosylation sites on the heavy chain. In addition, in IgA1 the hinge region is elongated (16 amino acid insertion rich in Pro, Ser and Thr) and modified with several (3 to 6) O−linked oligosaccharides. These sugar chains are generally small and heterogeneous and are likely to affect the structure adopted by the hinge region and in consequence the conformational variability of IgA1. This is important for the biological activity since the extended hinge in human IgA1 seems to afford a greater reach between its two antigen-binding sites, allowing this subclass to achieve high-avidity interaction with more distantly spaced antigens than other immunoglobulin isotypes (which might be the basis of the pronounced efficacy of IgA−based antibodies against bacterial pathogens). Moreover, the joining (J) chain in the dimeric IgA form is also N−glycosylated as is the “secretory component” (sc) of the secretory forms (sIgA1 and sIgA2).
To assess the impact of glycosylation on structure−function relationships of the various IgA formats, an expression platform is needed which allows the production of IgA variants bearing defined glycans. Recently, the tobacco−related species Nicotiana benthamiana has emerged as promising host for expression of recombinant glycoproteins with custom−made N− and O−glycan modifications (Strasser et al., 2014). Consequently, we hypothesize that functional IgA variants (IgA1, IgA2, dIgA1, dIgA2) with defined glycans can be generated in this expression platform and thus allow detailed characterization by a broad set of biochemical/physical techniques and functional assays. Such a project clearly needs an interdisciplinary approach involving faculty members from several BioToP areas. The present project will be headed by Richard STRASSER (expression platform, glycosylation engineering and characterization, functional assays) and will be performed in close cooperation with Christian OBINGER (spectroscopy, calorimetry) and Rupert TSCHELIESSNIG (small angle X−ray scattering).
Aims and methods.
This project aims to establish a robust expression platform for monomeric and dimeric IgA variants with homogeneous glycosylation profiles and to analyse the impact of glycosylation on structure, conformational and thermal stability as well as antigen binding and interaction with host receptors [e.g. FcαRI (CD89) or polymeric Ig receptor, pIgR].
Monomeric IgA1 and IgA2 variants of an anti−epidermal growth factor receptor (EGFR) antibody (e.g. cetuximab) will be transiently expressed in N. benthamiana plants using different expression vectors and subcellular targeting signals. The correct assembly, integrity and subcellular deposition of the recombinant IgAs will be carefully analysed to establish optimized expression conditions. The recombinant IgA antibodies will be purified and glycopeptides will be subjected to LC−ESI−MS analysis to determine the N−/O−glycan composition. Potentially beneficial glycoforms (e.g. lacking core fucose, high content of sialic acid) will subsequently be generated by expression in glycoengineered N. benthamiana (Strasser et al., 2008) and by co−expression of mammalian glycosylation enzymes and other proteins required for sialylated complex N-glycan (Castilho et al., 2010) and mucin−type O−glycan formation (Castilho et al., 2012). The capability of plants to produce functional dimeric IgA variants with defined glycosylation will be investigated by co−expression of the J chain.
The various formats (monomeric versus dimeric) and variants with defined glycosylation will be investigated concomitantly for their overall structure and homogeneity [size exclusion chromatography combined with multi-angle light scattering, circular dichroism (CD) spectroscopy], as well as conformational and thermal stability (CD and fluorescence spectroscopy, differential scanning calorimetry). For further structural evaluation small−angle X−ray scattering (SAXS) will be employed for evaluation of the overall shape and compactness of selected recombinant IgAs (Chromikova et al., 2015). Moreover, the effect of N−/O−glycosylation on proteolytic susceptibility will be determined by in vitro assays with IgA−specific proteases in a similar manner as previously reported for IgGs (Niemer et al., 2014). Functional characterisation of IgA glycoforms will be carried out e.g. by measurement of EGFR and Fcα−receptor binding.
Taken together, our study aims to establish a robust plant−based expression system for IgA glycoforms in order to systematically evaluate the impact of glycosylation on structure and function of IgA1 and IgA2 in both monomeric and dimeric forms.
Further collaborations within this thesis will include ALTMANN (mass spectrometry and glycan analysis), MACH (receptor binding assays, protease treatments), and STEINKELLNER (expression and purification of recombinant glycoproteins from plants).
Boross, P., Lohse, S., Nederend, M., Jansen, J.H., van Tetering, G., Dechant, M., Peipp, M., Royle, L., Liew, L.P., Boon, L., van Rooijen, N., Bleeker, W.K., Parren, P.W., van de Winkel, J.G., Valerius, T., Leusen, J.H. (2013) IgA EGFR antibodies mediate tumour killing in vivo. EMBO Mol. Med. 5, 1213-1226.
Castilho, A., Strasser, R., Stadlmann, J., Grass, J., Jez, J., Gattinger, P., Kunert, R., Quendler, H., Pabst, M., Leonard, R., Altmann, F., Steinkellner, H. (2010) In planta protein sialylation through overexpression of the respective mammalian pathway. J. Biol. Chem. 285, 15923-15930.
Castilho, A., Neumann, L., Daskalova, S., Mason, H. S., Steinkellner, H., Altmann, F., Strasser, R. (2012) Engineering of sialylated mucin-type O-glycosylation in plants. J. Biol. Chem. 287, 36518-36526.
Chromikova, V., Mader, A., Hofbauer, S., Goebl, C., Madl, T., Gach, J.S., Bauernfried, S., Furtmueller, P.G., Forthal, D.N., Mach, L., Obinger, C., Kunert, R. (2015) Introduction of germline residues improves the stability of anti-HIV mAb 2G12-IgM. Biochim. Biophys. Acta, in press.
Jefferis, R. (2009) Glycosylation as a strategy to improve antibody-based therapeutics. Nature Rev. Drug Discov. 8, 226–234.
Niemer, M., Mehofer, U., Torres Acosta, J.A., Verdianz, M., Henkel, T., Loos, A., Strasser, R., Maresch, D., Rademacher, T., Steinkellner, H., Mach, L. (2014) The human anti-HIV antibodies 2F5, 2G12 and PG9 differ in their susceptibility to proteolytic degradation: downregulation of endogenous serine and cysteine proteinase activities could improve antibody production in plant-based expression platforms. Biotechnol. J. 9, 493-500.
Strasser, R., Stadlmann, J., Schaehs, M., Stiegler, G., Quendler, H., Mach, L., Gloessl, J., Weterings, K., Pabst, M., Steinkellner, H. (2008) Generation of glyco-engineered Nicotiana benthamiana for the production of monoclonal antibodies with a homogeneous human-like N-glycan structure. Plant Biotechnol. J. 6, 392-402.
Strasser, R., Altmann, F., Steinkellner, H. (2014) Controlled glycosylation of plant-produced recombinant proteins. Curr. Opin. Biotechnol. 30, 95–100.
