Defined gp140 trimers as HIV vaccine
SUPERVISOR: RENATE KUNERT
Background.
The expression of adequate amounts of a recombinant protein is crucial to transfer a scientific idea into an applicable technology to continue with clinical trials. But cell biologists and technologists also need the interconnection with the field of protein chemistry and also virology to predict the utility of a sophisticated protein in a vaccination approach. Especially the human immunodeficiency virus (HIV) is a challenging pathogen for different reasons of limiting development of neutralizing antibodies through:
The success of recombinant mAb production has launched in the last decade with recombinant CHO clones expressing volumetric titres in the range of g/l. However, the demand for other and even more complicated proteins in terms of post translational modifications needs to be realized by defining a platform which includes cell line development and process optimization.
Aims and methods.
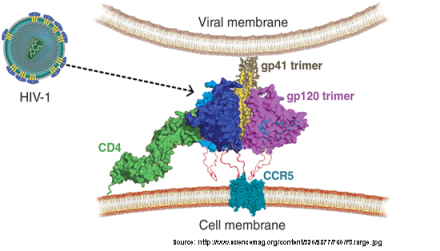
Definition of the optimal expression system for gp140 trimers and analysis of the protein
To mimic the naturally occurring envelope protein for the vaccination approach it is crucial that post translational modifications are executed correctly by the recombinant CHO cells. Beside other post-translational modifications the translated protein needs to be cleaved to form none covalently associated dimers to mimic the naturally occurring HIV envelope trimers (Moulard, M., and Decroly, E., 2000). Therefore the different CHO host cells will be tested for pro-protein converting enzyme activity to start with the best performing host. If necessary, proteases like furin, PC5 or PC7 (Liu et al., 2014) will be co-expressed to optimize the gp140 maturation process. For gene transfer we will apply BAC technology and introduce transgenes in the murine Rosa26 (Zboray et al, 2015) locus. Applicable methods for protein characterization are 1) Blue-native PAGE, 2) SEC, 3) stability testing in the presence of detergents (ionic detergent SDS, non-ionic detergents NP-40, Tween-20, Triton X-100) and accessibility of different anti HIV antibodies, 4) sedimentation equilibrium analysis of cleaved and uncleaved gp140s 5) incubation of gp140 with four domain sCD4 at room temperature for 30 min and then analysed by negative-stain EM.
Previous work has shown that monomeric forms of gp120 (the soluble part of the HIV envelope protein) are not sufficient to induce neutralizing antibodies in different prime-boost regimes to develop an HIV vaccine. Recent advances were realized by testing a trimeric form of gp140, an artificial but soluble variant of the gp160 precursor. Different approaches to stabilize the trimeric structure were already investigated, and in this project factors influencing the structure of gp140 trimers will be elucidated in more detail to develop new variants of cleaved and stabilized gp140 trimers.
In previous experiments we were able to express one variant of gp140 in up to 1000 mg/l titers in recombinant CHO cells. However the quality of the product was inhomogeneous regarding trimerization of the molecule and parts of the final product were lost during the downstream process. The combination of sophisticated new gp140 variants and the elaborated CHO expression system will define cell clones for fine tuning of process parameters (pH or temperature shift). Additionally, the cultivation medium will be optimized according to medium ingredients and supplements to provide a preferable environment for protein processing and the stabilization of the non-covalently linked trimeric structure of gp140.
Moulard, M., and Decroly, E. (2000). Maturation of HIV envelope glycoprotein precursors by cellular endoproteases. Biochim. Biophys. Acta BBA - Rev. Biomembr. 1469, 121–132.
Zboray, K., Sommeregger, W., Bogner, E., Gili, A., Sterovsky, T., Fauland, K., Grabner, B., Stiedl, P., Moll, H.P., Bauer, A., et al. (2015). Heterologous protein production using euchromatin-containing expression vectors in mammalian cells. Nucleic Acids Res. 43, e102
Liu, J., Jonebring, A., Hagström, J., Nyström, A.C., Lövgren, A. (2014) Improved expression of recombinant human factor IX by co-expression of GGCX, VKOR and furin. Protein J.; 33(2):174-83. doi: 10.1007/s10930-014-9550-5.
