Introduction of unnatural amino acids in recombinant proteins with the yeast Pichia pastoris
SUPERVISOR: DIETHARD MATTANOVICH
Background.
In vivo incorporation of unnatural amino acids (UAA) into proteins has been established more than a decade ago (Chin et al., 2003) with the aim of adding additional functions to proteins at specific sites. UAAs with chemical reactivity enable structurally defined conjugation of drugs to proteins (Kim et al., 2013b). Incorporation of UAAs makes typically use of orthogonal tRNAs complementary to a stop codon combined with an engineered aminoacyl-tRNA synthetase for this stop codon, and has among others been demonstrated to work in yeasts (Chen et al., 2007). Thus the incorporation of para-acetylphenylalanine into human serum albumin in P. pastoris was demonstrated (Young et al., 2009). The functional application of orthogonal pairs of E. coli tyrosyl- and leucyl-tRNA synthetase/tRNA pairs has already been successfully transferred to P. pastoris (Young et al., 2009), as the system could be used to incorporate 8 different unnatural amino acids in response to an amber codon. Further, a solvent-exposed glutamate residue on P. pastoris produced human serum albumin could be converted to p-acetylphenylalanine, which allowed an efficient conjugation of the modified protein with a therapeutic peptide via oxime ligation. Click chemistry is another option for conjugation of drugs to UAAs. Thus a proof principle of UAA incorporation has been established in P. pastoris however still far from a technological application. The aim of this thesis is to provide novel solutions to circumvent the major drawbacks and to develop a platform for the application of this technology to antibody-drug conjugate development.
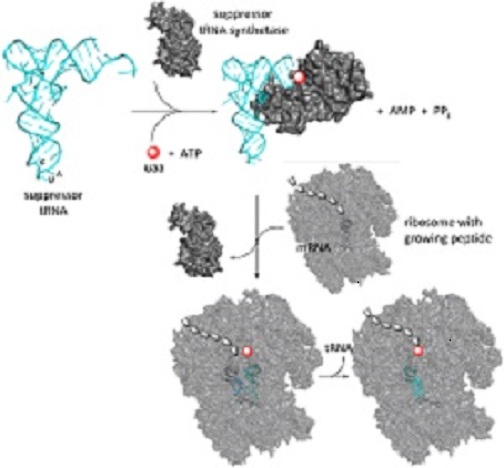
Aims and methods.
As a first benchmark experiment, an amber codon (UAG) suppressor vector will be designed containing the genes encoding the suppressor tRNA and the corresponding aminoacyl-tRNA synthetase. Different antibody fragments will be used as target proteins for UAA incorporation, testing different promoters for the best combination for protein overproduction and UAA incorporation. After purification, the composition of the modified protein will be verified with LC-MS/MS after tryptic digest. Other issues of UAA incorporation relate mainly to the availability of the UAA in the cell.
Incorporation of UAA into different antibody fragment formats will be tested, and the efficiency of the platform will be evaluated in bioreactor cultivation, measuring productivity, incorporation efficiency, protein homogeneity and the efficiency of drug coupling.
Chen, S., P. G. Schultz, and A. Brock, 2007, An improved system for the generation and analysis of mutant proteins containing unnatural amino acids in Saccharomyces cerevisiae: J Mol Biol, v. 371, p. 112-22.
Chin, J. W., T. A. Cropp, J. C. Anderson, M. Mukherji, Z. Zhang, and P. G. Schultz, 2003, An expanded eukaryotic genetic code: Science, v. 301, p. 964-7.
Young, T. S., I. Ahmad, A. Brock, and P. G. Schultz, 2009, Expanding the genetic repertoire of the methylotrophic yeast Pichia pastoris: Biochemistry, v. 48, p. 2643-53.
