Changing the substrate specifity of lytic polysaccharide monooxygenase
SUPERVISOR: CHRIS OOSTENBRINK
Background.
The need for renewable energy is growing rapidly and biofuel derived from lignocellulose is an attractive alternative to fossil-based fuels. Notwithstanding, the visionary green energy concept is challenged by the recalcitrant nature of the major component cellulose, which makes bioconversion to fermentable saccharides problematic and costly. (Quinlan et al. 2011). Enzymatic degradation of recalcitrant polysaccharides occurs through the synergistic action of hydrolytic enzymes that have complementary activities. Endo-acting hydrolases make random scissions, whereas exo-acting processive hydrolases mainly target chain ends.
However, a new enzyme family targeting recalcitrant polysaccharides has been identified, namely the lytic polysaccharide monooxygenase (LPMO). It catalyzes oxidative cleavage of glycosidic bonds that results in oxidation of the C1 or C4 carbon. This reaction involves molecular oxygen and a copper atom that is coordinated by two conserved histidines. (Aachmann et al., 2012; Vermaas et al., 2015) The precise mechanism of the monooxygenation reaction is hereto unknown, but it involves reduction of the copper center of LPMO by a reducing agent such as ascorbic acid or by a co-secreted enzyme such as cellobiose dehydrogenase (CDH) and the formation of a reactive oxygen species to initiate substrate hydroxylation, either by a superoxo or an oxyl mechanism (Borisova et al., 2015; Kim et al., 2014; Li et al., 2012) To gain a better insight/understanding of the catalytic mechanism and the relationship between active site and substrate, it is necessary to modify the active site and to test different possible/alternative substrates.
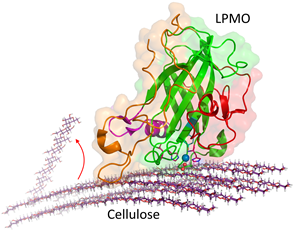
As a first step into this endeavour, the full structure of the LPMO from N. crassa was elucidated. The structure and function of complex macromolecular entities like proteins have been studied by molecular simulation and led to insight into the molecular mechanisms that underlie experimental observations (Karplus and Kuriyan, 2005; van Gunsteren et al., 2006) Sampling involves the generation of a sufficiently large amount of molecular structures to describe the orientations and conformations that may be obtained from a variety of search techniques, like systematic searches, molecular dynamics or Monte Carlo simulations. Experimental proof will be obtained from fast-kinetic and calorimetric techniques in combination with rational protein engineering.
Aims and methods.
In this project, we will study the catalytic mechanism and the substrate binding of LPMO. A starting point for these studies is N. crassa LPMO or other LPMOs with published structural data (Hemsworth et al., 2013). The crystal structure of the LPMO shows that the copper is centered in a flat surface, which is facing the surface of cellulose during catalysis.
It will be of particular interest to determine whether alternative substrates and substrate binding to the LPMO active site are feasible by using substrate docking and molecular modelling simulations. By constructing specific mutations to influence the substrate binding site of the LPMO, the suggested function of the amino acids will validated experimentally. Amino acids identified as important for substrate binding will be mutated to prove or disprove their importance.
Enzyme variants will be produced in Pichia pastoris or Trichoderma reesei and studied by biochemical, bioelectrochemical and biophysical methods including rapid kinetics as well as EPR and isothermal calorimetry.
Aachmann, F.L., Sorlie, M., Skjak-Bræk, G., Eijsink, V.G.H., Vaaje-Kolstad, G. (2012) NMR structure of a lytic polysaccharide monooxygenase provides insight into copper binding, protein dynamics, and substrate interactions. Proc. Natl. Acad. Sci. 109, 18779–18784. doi:10.1073/pnas.1208822109
Borisova, A.S., Isaksen, T., Dimarogona, M., Kognole, A.A., Mathiesen, G., Varnai, A., Rohr, A.K., Payne, C.M., Sorlie, M., Sandgren, M., Eijsink, V.G.H. (2015) Structural and Functional Characterization of a Lytic Polysaccharide Monooxygenase with Broad Substrate Specificity. J. Biol. Chem. 290, 22955–22969. doi:10.1074/jbc.M115.660183
Hemsworth, G.R., Davies, G.J., Walton, P.H. (2013) Recent insights into copper-containing lytic polysaccharide mono-oxygenases. Curr. Opin. Struct. Biol., Protein-carbohydrate interactions. Biophysical methods 23, 660–668. doi:10.1016/j.sbi.2013.05.006
Karplus, M., Kuriyan, J. (2005) Molecular dynamics and protein function. Proc. Natl. Acad. Sci. U. S. A. 102, 6679–6685. doi:10.1073/pnas.0408930102
Kim, S., Stahlberg, J., Sandgren, M., Paton, R.S., Beckham, G.T. (2014) Quantum mechanical calculations suggest that lytic polysaccharide monooxygenases use a copper-oxyl, oxygen-rebound mechanism. Proc. Natl. Acad. Sci. 111, 149–154. doi:10.1073/pnas.1316609111
Li, X., Beeson IV, W.T., Phillips, C.M., Marletta, M.A., Cate, J.H.D. (2012) Structural Basis for Substrate Targeting and Catalysis by Fungal Polysaccharide Monooxygenases. Structure 20, 1051–1061. doi:10.1016/j.str.2012.04.002
van Gunsteren, W.F., Bakowies, D., Baron, R., Chandrasekhar, I., Christen, M., Daura, X., Gee, P., Geerke, D.P., Glaettli, A., Huenenberger, P.H., Kastenholz, M.A., Oostenbrink, C., Schenk, M., Trzesniak, D., van der Vegt, N.F.A., Yu, H.B. (2006) Biomolecular Modeling: Goals, Problems, Perspectives. Angew. Chem. Int. Ed. 45, 4064–4092. doi:10.1002/anie.200502655
Vermaas, J.V., Crowley, M.F., Beckham, G.T., Payne, C.M. (2015) Effects of Lytic Polysaccharide Monooxygenase Oxidation on Cellulose Structure and Binding of Oxidized Cellulose Oligomers to Cellulases. J. Phys. Chem. B 119, 6129–6143. doi:10.1021/acs.jpcb.5b00778
