Understanding molecular transport processes for improved metabolic engineering approaches
SUPERVISOR: MICHAEL SAUER
Background.
Membrane transport proteins play a key role in many metabolic pathways within cells. They either actively or passively transport ions, metabolites or proteins across the membrane in or out of the cell or from one cellular compartment to another. A better understanding of the membrane processes can be applied in metabolic engineering to optimize metabolic pathways for the production of valuable metabolites (Steiger et al., 2016).
However, the main problem in the studies of membrane proteins lies within their hydrophobic characteristics and their lipid membrane-dependent functionality (Seddon et al., 2004). Membrane proteins are investigated in detergent environments or are reconstituted into lipid based platforms such as liposomes or nanodiscs (Shen et al., 2013). As liposomes can be used to simulate compartments, they are ideal investigation tools to study membrane protein functionalities and their transport processes. Although there exists difficulties concerning protein solubilization and orientation, successful incorporation of proteins in liposomes, namely proteoliposomes, and subsequent characterization of the transporter processes have been described (Palmieri et al., 1997). However, this technology is far from routinely being used.
Aims and methods.
The main objective of this study is the incorporation of fully functional transport proteins into lipid based platforms for a comprehensive functional in vitro characterization.
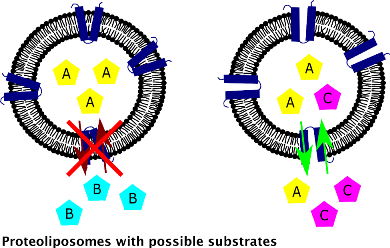
Firstly, investigations will focus on heterologous production of selected mitochondrial membrane transport proteins. After optimizing the expression and an appropriate purification process of the membrane proteins, the following work will focus on finding detergent and lipid based conditions for the transporters to ensure their functionality. In the further working steps, the functional transporters would be finally incorporated into liposomes. Subsequently, transport assays with possible substrates will be performed to elucidate the functionality and specificity of these proteins. These findings can then be used for the construction of tailor-made metabolic pathways.
CPalmieri, L., Lasorsa, F.M., De Palma, A., Palmieri, F., Runswick, M.J., Walker, J.E., 1997. Identification of the yeast ACR1 gene product as a succinate-fumarate transporter essential for growth on ethanol or acetate. FEBS Lett. 417, 114–118.
Seddon, A.M., Curnow, P., Booth, P.J., 2004. Membrane proteins, lipids and detergents: Not just a soap opera. Biochim. Biophys. Acta - Biomembr. 1666, 105–117.
Shen, H.H., Lithgow, T., Martin, L.L., 2013. Reconstitution of membrane proteins into model membranes: Seeking better ways to retain protein activities. Int. J. Mol. Sci. 14, 1589–1607.
Steiger, M.G., Punt, P.J., Ram, A.F.J., Mattanovich, D., Sauer, M., 2016. Characterizing MttA as a mitochondrial cis-aconitic acid transporter by metabolic engineering. Metab. Eng. 35, 95–104.
