Immunoglobulin E (IgE) and glycosylation
SUPERVISOR: HERTA STEINKELLNER
Background.
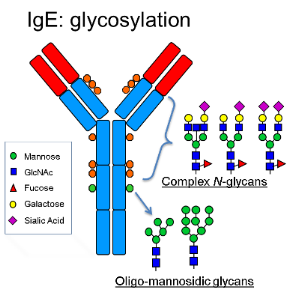
Immunoglobulins are the most abundant proteins in human serum. Like other plasma proteins they are heterogeneously glycosylated reflecting their multiple functions. Antibodies of the IgE class are key mediators of the allergic response and confer protection against parasitic infections. Therapeutic antibodies, mainly of the IgG class, have revolutionized modern medicine. Recent studies show that IgE exhibit modalities which allows them to be used as a novel anti-cancer modality (Karagiannis 2012). An interesting feature of IgE antibodies is their complex glycosylation pattern, approx. 15 % of the molecular weight refers to carbohydrates. To date, how this posttranslational modification contributes to the function of IgEs remains unexplored. However, previous studies indicated that carbohydrates play a structural and functional role for all immunoglobulins (Zauner 2013).
Aims and methods.
In this proposal we aim to produce IgE and a series of glycosylation variants thereof and determine their in vitro and in vivo activities. For the production a plant based expression system, developed in the applicant`s lab, allowing the generation of pre-specified human glycoforms on demand, will be used (Strasser et al., 2014). Selected monoclonal IgE (i.e. pharmaceutically interestingly IgE against epidermal growth factor receptor 2, HER2/neu, (Karagiannis et al., 2009), will be expressed. The synthesis of various glycoforms will be rationally designed according to their natural presence in serum. This collection of glyco-variants will be subjected detailed structural analyses (HPLC, MALDI-TOF MS/MS in cooperation with PI Altmann). Subsequent biochemical and biophysical characterization (e.g. oligomerisation status, thermal stability) will be carried out to reveal possible glycan associated modifications. In order to elucidate the impact of glycans to functional activities in vitro and in vivo studies (i.e. antigen-, receptor binding, tumor killing potency) will be performed. Finally, experimental data will be evaluated in the context of Computer based simulation and modelling of IgE with the aim to rationalise experimental data (in cooperation with PI Oostenbrink).
Karagiannis SN, Josephs DH, Karagiannis P, et al., (2012). Recombinant IgE antibodies for passive immunotherapy of solid tumours: from concept towards clinical application. Cancer Immunol Immunother. 61:1547-64.
Karagiannis P, Singer J, Hunt J, Gan SK et al., (2009) Characterisation of an engineered trastuzumab IgE antibody and effector cell mechanisms targeting HER2/neu-positive tumour cells. Cancer Immunol Immunother. 58:915-30
Strasser R, Altmann F, Steinkellner H. (2014) Controlled glycosylation of plant-produced recombinant proteins. Curr Opin Biotechnol. 30:95-100.
Zauner G, Selman MH, Bondt A, Rombouts Y, Blank D, Deelder AM, Wuhrer M. (2013) Glycoproteomic analysis of antibodies. Mol Cell Proteomics 12:856–865.
