Identification of Lewis a containing glycoproteins from Arabidopsis thaliana and characterization of their involvement in pathogen defence
SUPERVISOR: RICHARD STRASSER
Background.
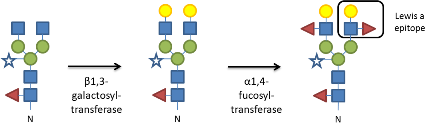
The attachment of an oligosaccharide-precursor to certain asparagine residues present on newly synthesized polypeptide chains, termed N-glycosylation, is one of the most abundant protein modifications. The wide variety of structures found among distinct organisms is the result of differential processing of the attached sugars by the action of specific glycosylases and glycosyltransferases. In metazoans, the terminal sugar residues present on complex N-glycans regulate a large number of diverse biological processes by modulating receptor-ligand interactions, for example during removal of glycoproteins from circulation or during receptor signaling. In addition to the endogenous recognition functions, cell surface glycoconjugates on mammalian cells are also sites for pathogen recognition and attachment. By contrast, surprisingly little is known about the function of complex N-glycan modifications in plants (Strasser, 2014). The only known outer chain elongation of complex N-glycans is the attachment of β1,3-galactose- and α1,4-fucose-residues to terminal N-acetylgucosamine, forming a trisaccharide known as the Lewis a epitope (Strasser et al., 2007). These modifications take place in the trans-Golgi and are carried out by the sequential action of β1,3-galactosyltransferase and α1,4-fucosyltransferase. The Lewis a epitope was detected primarily on soluble and membrane-bound extracellular glycoproteins from different plant species, but the proteins carrying this glycan and the biological function of Lewis a structures are largely unknown.
Aims and methods.
(i) Identification of Lewis a containing proteins from Arabidopsis organs like stems, siliques and trichomes
Lewis a-containing glycoproteins will be isolated from Arabidopsis thaliana via affinity purification. Isolated proteins will be identified via LC-ESI-MS analysis and used for further experiments, such as generation of knockout- and overexpression-lines; selected target proteins will furthermore be characterized in order to decipher the unknown function of Lewis a-structures in plants.
(ii) Analysis of F-box-Nictaba binding to Lewis a containing proteins
Previous studies showed that the stress-inducible F-box-Nictaba protein from Arabidopsis thaliana binds to N-acetyllactosamine- and Lewis a-structures in a glycan array experiment and is coexpressed together with α1,4-fucosyltransferase and β1,3-galactosyltransferase (Stefanowicz et al., 2012). Overexpression lines of the F-box-Nictaba gene were furthermore less susceptible to pathogen infection (Stefanowicz et al., 2016). Binding of F-box-Nictaba to different Lewis a-containing glycoproteins will be investigated.
(iii) Investigation of the role of complex N-glycosylation and Lewis a –structures for pathogen infection
In order to study a possible involvement of Lewis a structures in pathogen defence, Arabidopsis Lewis a knockout and overexpression lines will be subjected to pathogen infection assays.
Stefanowicz K., Lannoo N., Proost P., Van Damme E. J. M. (2012). Arabidopsis F-box protein containing a Nictaba-related lectin domain interacts with N-acetyllactosamine structures. FEBS Open Bio 2, 151–158.
Stefanowicz K., Lannoo N., Zhao Y., Eggermont L., Van Hove J., Al Atalah B., et al. . (2016). Glycan-binding F-box protein from Arabidopsis thaliana protects plants from Pseudomonas syringae infection. BMC Plant Biol. 16:213.
Strasser R., Bondili J., Vavra U., Schoberer J., Svoboda B., Glössl J., et al. (2007). A unique beta1,3-galactosyltransferase is indispensable for the biosynthesis of N-glycans containing Lewis a structures in Arabidopsis thaliana. Plant Cell 19 2278–2292
Strasser R., Biological significance of complex N‐glycans in plants and their impact on plant physiology. Front. Plant Sci. 2014, 5, 363.
