Inducible regulation of baculovirus genes involved in secretion and virus budding
SUPERVISOR: REINGARD GRABHERR
Background.
Virus-like particles (VLPs) are empty shell protein structures that are used as effective and safe vaccines for animals and humans. VLPs are non-infectious and mimic the outer surface of a virus. The baculovirus insect cell expression platform is often the method of choice for the production of enveloped as well as non-enveloped VLPs (Grabherr and Krammer, 2010). MERS-Coronavirus VLPs (Wang et al., 2017), human enterovirus VLPs (Zhao et al., 2017), porcine epidemic diarrhea virus VLPs (Wang et al., 2017), chicken influenza virus VLPs (Wu et al., 2017) and Marburg virus VLPs (Weiwei et al., 2017) are just a few recent examples for insect cell produced VLP vaccines. In the case of enveloped particles, the challenge lies in down-stream processing since the initial harvest is a mixture of VLPs, budded baculoviruses, exosomes deriving from cellular exocytosis and microvesicles that bud from the plasma membrane (Koppen et al., 2011; Steppert et al., 2017).
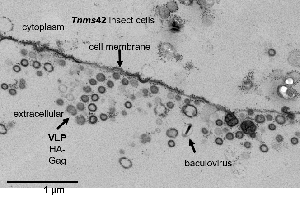
In order to improve the product quality and facilitate down-stream processing, virus and cell engineering tools are needed to down-regulate the production of un-wanted particle components. In the past, production strategies have been tested that avoid the budding of baculovirus particles into the supernatant. The use of mammalian cell lines (Thompson et al., 2015) or non-budding baculoviruses for infection of insect cells (Chaves et al., 2018) was so far not feasible due to a massive loss of yield. The strategy to delete genes from the baculovirus genome that are responsible for budding is possible, however requires helper cell lines providing the missing gene, which often is toxic to the cells or at least hampers cell growth (Chaves et al., 2018; Marek et al., 2011). Yet, there exists no efficient strategy to control the product composition, yield and quality by genetic engineering of the baculovirus expression vector.
Aims and methods.
Our goal is to construct an expression system that can be used to control baculovirus budding during the infectious cycle and at the same time allows for VLP production. In the past we have applied microRNA based strategies to knock down specific baculovirus genes, however this was only partly efficient (data not shown). Alternatively, CRISPR/Cas9 technology was recently developed for insect cells (Mabashi-Asazuma and Jarvis, 2017; Chavez-Pena and Kamen, 2018), however not yet for genes on the baculovirus genome. Both these methods will be established and optimized, so that upon baculovirus infection, genes that are involved in budding should be either down-regulated (antisense-RNA) or deleted (CRISPR/Cas9), either one single gene or several in combination. One critical aspect is that the target gene does not interfere with virus replication and down-regulation/deletion still allows unrestricted productivity.
First targets will be VP80, which is part of the baculovirus capsid structure and was shown to be essential for virus budding (Marek et al., 2011) and ac75, a protein required for nuclear egress of the virus capsid (Shi et al., 2018). Downregulation of baculovirus budding is expected to decrease or abolish secondary infection of cells. Therefore, initial infection must be done at high multiplicity of infection and under optimized conditions. We have seen that some insect cells tolerate alkaline pH, which leads to a highly increased uptake of baculoviruses by endocytosis. This and similar measures will be tested and optimized. Process performance, robustness, yield of product and reduction of baculovirus budding will be determined. The overall goal is to develop a set of genetic tools to engineer baculovirus vectors for inducible tuning (downregulation) of baculovirus budding as well as other genes that are involved in extracellular particle production. All approaches will be tested in small and large scale, fed batch and continuous production processes.
Collaborations within this thesis include JUNGBAUER (particle preparation) and VORAUER-UHL (analytics of particles).
Burgstaller, D., Klausberger, M., Kramberger, P., Tover, A., Berger, E., Nöbauer, K., Razzazi-Fazeli, E., Jungbauer, A. (2017) Separation of HIV-1 gag virus-like particles from vesicular particles impurities by hydroxyl-functionalized monoliths. J. Sep. Sci. 40, 979-990. doi: 10.1002/jssc.201600765.
Chaves, L.C.S., Ribeiro, B.M., Blissard, G.W. (2018) Production of GP64-free virus-like particles from baculovirus-infected insect cells. J. Gen. Virol. 99. 265-274. doi: 10.1099jgv.0.001002
Chavez-Pena, C. and Kamen, A. (2018) RNA interference technology to improve the baculovirus-insect cell expression system. Biotechnol. Adv. 36, 443-451. doi: 10.1016/j.biotechadv.2018.01.008
Gai, W., Xuexing, Z., Chong, W., Yongkun, Z., Qi, W., Hualei, W., Gary, W., Ying, X., Haijun, W., Zengguo, C., Na, F., Hang, C., Tiecheng, W., Yuwei, G., Junjie, S., Songtao, Y., Xianzhu, X. (2017) Marburg virus-like particles produced in insect cells induce neutralizing antibodies in rhesus macaques. J. Med. Virol. 89, 2069-2074. doi: 10.1002/jmv.24832
Koppen, T., Weckmann, A., Müller, S., Staubach, S., Bloch, W., Dohmen, R.J., Schwientek, T. (2011) Proteomics analyses of microvesicles released by Drosophila Kc167 and S2 cells. Proteomics 11, 4397-4410. doi: 10.1002/pmic.201000774
Krammer, F. and Grabherr, R. (2010) Alternative Influenza Vaccines Made by Insect Cells. Trends Mol. Med. 16, 313-320. doi:10.1016/jmolmed.2010.05.002
Mabashi-Asazuma, H. and Jarvis, D.L. (2017) CRISPR-Cas9 vectors for genome editing and host engineering in the baculovirus–insect cell system. Proc. Nat. Acad. Sci. USA 114, 9068-9073. doi: 10.1073/pnas.1705836114
Marek, M., vanOers, M.M., Devaraj, F.F., Vlak, J.M., Merten, O.-W. (2011) Engineering of baculovirus vectors for the manufacture of virion-free biopharmaceuticals. Biotechnol. Bioeng. 108, 1056-1067. doi: 10.1002/bit.23028
Shi, A., Hu, Z., Wang, Y., Wu, W., Yang, K. (2018) Autographa californica nuclearpolyhedrosisvirus ac75 is required for the nuclear egress of nuclearcapsids and intranuclear microvesicle formation. J. Virology 92, e01509-17. doi: 10.1128/JVI.01509-17
Thompson, C.M.,Petiot, E., Mullick, A., Aucoin, M.G., Henry, O., Kamen, A.A. (2015) Critical assessment of influenza VLP production in Sf9 and HEK293 expression systems. BMC Biotechnol. 15, 19-47. doi: 10.1186/s12896-015-0152-x
Wang, A., Gu, L., Wu, S., Zhu, A. (2018) Duck hepatitis A structural proteins expressed in insect cells self-assemble into virus-like particles with strong immunogenicity in ducklings. Vet. Microbiol. 215, 23-28. doi: 10.1016/j.metmic.2017.12.020
Wang, C., Yan, F., Zheng, X., Wang, H., Jin. H., Wang, C., Zhao, Y., Feng, N., Wang, T., Gao, Y., Yang, S., Xia, X (2017) Porcine epidemic diarrhea virus virue-like particles produced in insect cells induce specific immune response in mice. Virus Genes 53, 548-554. doi: 10,1007/s11262-017-1450-2
Wang, C., Zheng, X., Gai, W., Zhao, Y., Wang, H., Wang, H., Feng, N., Chi, H., Qiu, B., Li, N., Wang, T., Gao, Y., Yang, S., Xia, X. (2017) MERS-CoV virus-like particles produced in insect cells induce specific and cellular immunity in rhesus macaques. Oncotarget 8, 12686-12694. doi: 10.18632/oncotarget.8475
Wu, P., Lu, J., Zhang, X., Mei, M. Feng, L., Peng, D., Hou, J., Kang, S.M., Liu, X., Tang, Y. (2017) Single dose of consensus hemagglutinin-based virus-like particles vaccine protects chickens against divergent H5 subtype influenza virus. Front. Immunol. 8, 1649. doi: 10.3389/fimmu.2017.01649
Zhao, D., Sun, B., Sun, S., Fu, B. et al. (2017) Characterisation of a human enterovirus71 virus-like particles used for vaccine strategies. PLoS One 12, e0181182. doi: 10.1371/ journal.pone.0181182
