Extracellular vesicles as drug-targeting vehicle
SUPERVISOR: JOHANNES GRILLARI
Background.
Extracellular vesicles (EVs) of mesenchymal stem cells, endothelial cells or even iPS cells are among the promises of novel drug delivery systems (Hromada et al., 2017; Giebel et al., 2016), especially for oligonucleotide drugs. EVs have been described to be secreted by most if not all cell types, to travel through the circulation and to be taken up by recipient cells, where their cargo consisting of a cocktail of proteins, mRNAs and ncRNAs alters the behaviour of the recipient cells – in a way that might be even considered similar to hormones or cytokines.
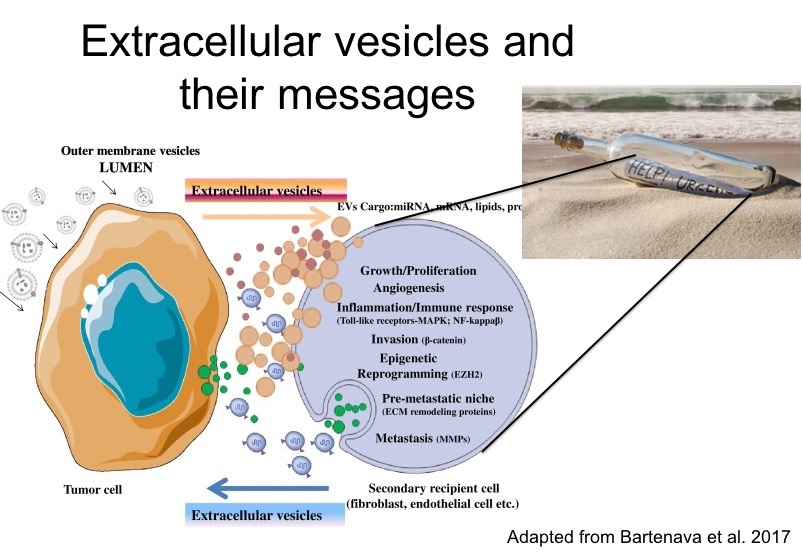
The specificity of the recipient cell targeting in vivo is understood in a limited way only, however, it is clear that receptor mediated endocytotic uptake, as well as membrane fusion after getting cell and vesicle membrane in close proximity are among various mechanisms that have been described. EVs do qualify as drug delivery system, as they can be loaded with different drugs in vitro (El Andaloussi et al. 2013), although they also can have detrimental effects when from senescent cells (Weilner et al., 2016a; Weilner et al., 2016b; Terlecki-Zaniewicz et al., 2018).
We have recently established recombinant EVs using Snorkel-tagged CD81 and CD63. Indeed, EVs from cells overexpressing these constructs can be isolated by single step affinity chromatography and eluted using prescission protease using a cleavage site integrated in the genetic construct.
Aims and methods.
In order to use EVs as a specific drug delivery vehicle, we will here (i) generate antibody-tagged EVs; (ii) produce sufficient amounts for targeting assays in vitro and in vivo; and (iii) proof the uptake of EVS in target cells. To this end, the Snorkel tag will be genetically fused to single chain antibodies recognizing EGFR (e.g. as scvAb in Akbari et al., 2016) as a model anti-tumor target (RÜKER). Then human telomerase immortalized mesenchymal stem cell lines as available in the lab (Wolbank et al., 2009) will be stably transfected with the respective construct and recombinant expression on the surface of EVs will be confirmed using immunogold labelling electron microscopy (STÖGER), Western blotting for presence of EV markers and absence of cytoplasmic markers, as well as immunofluorescence and nanoparticle tracking analysis. In order to produce sufficient EV amounts, cells will be cultivated in a simple hollow-fiber reactor (FibreCell systems) and harvested from media after removal of contaminating EVs present in fetal calf serum by ultracentrifugation.
The purified (JUNGBAUER), characterized resulting EVs will be used to load cytotoxic siRNA by lipofection as already established in the lab, exposed to EGFR-positive tumor cell lines and tested for cell viability. To assess the influence of EGFR-mediated binding to the total uptake of EVs, siRNA against EGFR will be used and compared to non-targeting control siRNA transfected cells.
Finally, the recombinant and siRNA and/or Cre mRNA loaded EVs will be tested in an EGFR positive tumor xenotransplant mouse model that is available at our collaboration partner Petra Heffeter, Vienna. The target tumor cells and CrispR/Cas9 EGFR deleted control cells will be stably transfected with a stop lox cassette in front of a luciferase reporter. By uptake of the EVs into EGFR-positive vs ko cells, Cre mRNA will be translated and remove the stop lox cassette. Thus, the targeting efficiency of the EVs and the contribution of the EGFR targeting can be quantitated. This will contribute to establish EVs as potential drug targeting vehicle.
Collaborations within this thesis will include JUNGBAUER (vesicle isolation), MACH (EV trafficking), STÖGER (ultrastructure analysis of EVs) and RÜKER (protein engineering)
Akbari, B., Farajnia, S., Zarghami, N., Mahdieh, N., Rahmati, M., Khosroshahi, S.A., Rahbarnia, L. (2016) Design, expression and evaluation of a novel humanized single chain antibody against epidermal growth factor receptor (EGFR). Protein Expr. Purif. 127, 8-15. doi: 10.1016/j.pep.2016.06.001.
El Andaloussi, S., Lakhal, S., Mäger, I., Wood, M.J.A. (2013) Exosomes for targeted siRNA delivery across biological barriers. Adv. Drug Deliv. Rev. 65, 391-397. doi: 10.1016/ j.addr.2012.08.008
Hromada, C., Mühleder, S., Grillari, J., Redl, H., Holnthoner, W. (2017) Endothelial extracellular vesicles-promises and challenges. Front. Physiol. 8, 275. doi: 10.3389/ fphys.2017.00275
Weilner, S., Keider, V., Winter, M., Harreither, E., Salzer, B., Weiss, F., Schraml, E., Messner, P., Pietschmann, P., Hildner, F., Gabriel, C., Redl, H., Grillari-Voglauer, R., Grillari, J. (2016a) Vesicular Galectin-3 levels decrease with donor age and contribute to the reduced osteo-inductive potential of human plasma derived extracellular vesicles. Aging (Albany NY) 8, 16-33. Erratum in: Aging (Albany NY). 8, 1156-1157. doi: 10.18632/aging.100865
Weilner, S., Schraml, E., Wieser, M., Messner, P., Schneider, K., Wassermann, K., Micutkova, L., Fortschegger, K., Maier, A.B., Westendorp, R., Resch, H., Wolbank, S., Redl, H., Jansen-Dürr, P., Pietschmann, P., Grillari-Voglauer, R., Grillari, J. (2016b) Secreted microvesicular miR-31 inhibits osteogenic differentiation of mesenchymal stem cells. Aging Cell. 15, 744-754. doi: 10.1111/acel.12484
Wolbank, S., Stadler, G., Peterbauer, A., Gillich, A., Karbiener, M., Streubel, B., Wieser, M., Katinger, H., van Griensven, M., Redl, H., Gabriel, C., Grillari, J., Grillari-Voglauer, R. (2009) Telomerase immortalized human amnion- and adipose-derived mesenchymal stem cells: maintenance of differentiation and immunomodulatory characteristics. Tissue Eng. Part A 15, 1843-1854. doi: 10.1089/ten.tea.2008.0205
Terlecki-Zaniewicz, L., Lämmermann, I., Latreille, J., Bobbili, M.R., Pils, V., Schosserer, M., Weinmüllner, R., Dellago, H., Skalicky, S., Pum, D., Higareda Almaraz, J.C., Scheideler, M., Morizot, F., Hackl, M., Gruber, F., Grillari, J. (2018) Extracellular vesicles and their miRNA cargo are anti-apoptotic members of the SASP. Aging, doi:10.18632/aging.101452
Yáñez-Mó, M. et al. (2015) Biological properties of extracellular vesicles and their physiological functions. J. Extracell. Vesicles 4, 27066. doi: 10.3402/jev.v4.27066
