Oxygen reactivity in the flavin-dependent enzyme pyranose oxidase
PRINCIPAL INVESTIGATOR: DIETMAR HALTRICH
Background.
Pyranose oxidase (POx; pyranose:oxygen 2-oxidoreductase; EC 1.1.3.10) is a member of the GMC family of FAD-dependent oxidoreductases and catalyzes the regioselective oxidation of aldopyranoses at position C2. During its first half-reaction, FAD is reduced by the sugar to FADH2 and re-oxidized in the second half-reaction by reducing molecular oxygen to H2O2. Alternative electron acceptors, including quinones, radicals or chelated metal ions, show significant and in some cases even higher activity than O2. Hence, the flavin-dependent enzyme POx shows both oxidase and dehydrogenase activity. Flavins are among a limited number of organic and inorganic biocatalysts that can effectively reduce oxygen (O2).
In solution the reaction of reduced flavin with O2 is autocatalytic but very slow, primarily because the initial single electron transfer from the singlet flavin hydroquinone to the triplet O2 is catalyzed poorly. In flavin-dependent enzymes, O2 reduction can be accelerated by 3–4 orders of magnitude or, alternatively, be abated, indicating that the protein microenvironment controls and modulates flavin reactivity (Massey, 1994; Chaiyen et al., 2012). It was suggested that amongst others a positive charge in the vicinity of the isoalloxazine, a hydrophobic site close to the flavin C(4a) as well as accessibility of the active site through tunnels and channels may be important aspects for oxygen reactivity of flavin-dependent enzymes, yet no general pattern can be given as to the positive or negative modulation of this reactivity with oxygen.
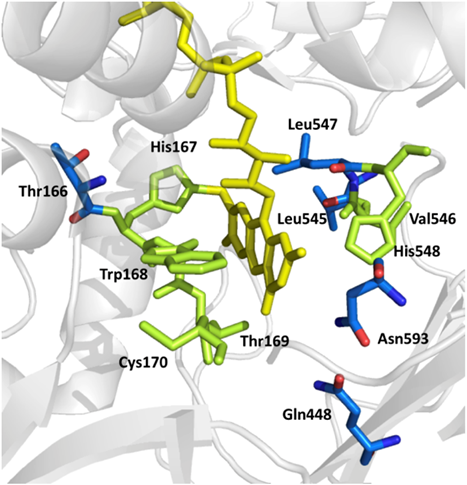
In a previous BioToP project (project Dagmar BRUGGER) we studied oxygen reactivity of POx and how it is affected by amino acid side chains in the vicinity of the isoalloxazine. To this end we applied site-saturation mutagenesis on a set of eleven amino acids around the active site of POx from Trametes multicolor. Variants T166R, Q448H, L545C, L547R and N593C were found to show decreased or almost completely abolished activity with oxygen as judged from their apparent steady-state constants and bioelectrochemical measurements, while activity with the alternative electron acceptors DCIP, 1,4-benzoquinone (1,4-BQ) and ferricenium ion (Fc+) was maintained (Brugger et al., 2014; Brugger et al., 2016).
Aims and methods.
In this project we will study the above-mentioned variants showing altered oxygen reactivity in detail, both biochemically and structurally. In addition, we will create new variants by combining interesting mutations at these different positions (or other positions around the isoalloxazine) in order to gain more insight into the structure/function relationship of flavoenzymes with respect to their reactivity with oxygen. This will be done by using CASTing (combinatorial active-site saturation test; Reetz et al., 2005), which allows the combination of random mutations in spatially close amino acids, together with a novel cell-based assay for high throughput screening of oxidoreductases, which was developed in an ongoing BioToP project (project Peter HERZOG). This random evolution of POx, extending the previous studies, will be preformed in cooperation with PETERBAUER.
So far, we only determined the apparent steady-state constants of the above-mentioned variants for O2, DCIP, 1,4-BQ and Fc+, with glucose as saturating substrate (Brugger et al., 2014). These previous studies showed altered oxidase activity combined with preserved dehydrogenase activity, but did not give any indication as to which of the two half-reactions of POx is affected by the amino acid exchanges in the active site. Hence we will use rapid kinetic experiments to elucidate the effect on the individual half reactions (FAD reduction and FADH2 re-oxidation) in detail. Apart from determining the rate constants using stopped flow spectrometry, we will look at different spectroscopic properties of the variants (to test whether the overall fold is intact; equipment at the BOKU Core Facility Biomolecular and Cellular Analysis together with OBINGER), bioelectrochemical properties (redox potential of the FAD, together with LUDWIG) as well as the thermodynamic stability of these variants. The structure of selected variants will be solved by X-ray crystallography together with external partners (Hallberg et al., 2004; Kujawa et al., 2006), and the positioning of O2 in these variants will be modeled (together with OOSTENBRINK) to gain information about e.g. the distance to the C4(a) to understand why oxygen reactivity is modified.
Collaborations within this thesis will include LUDWIG (electrochemistry), OBINGER (rapid kinetics, spectroscopic methods), OOSTENBRINK (molecular modelling and simulations), and PETERBAUER (enzyme evolution).
Brugger, D., Krondorfer, I., Shelswell, C., Huber-Dittes, B., Haltrich, D., Peterbauer, C.K. (2014) Engineering pyranose 2-oxidase for modified oxygen reactivity. PLoS One 9:e109242. doi: 10.1371/journal.pone.0109242
Brugger, D., Sützl, L., Zahma, K., Haltrich, D., Peterbauer, C.K., Stoica, L. (2016) Electrochemical characterization of the pyranose 2-oxidase variant N593C shows a complete loss of the oxidase function with full preservation of substrate (dehydrogenase) activity. Phys. Chem. Chem. Phys. 18, 32072-32077. doi: 10.1039/c6cp06009a
Chaiyen, P., Fraije, M.W., Mattevi, A. (2012) The enigmatic reaction of flavins with oxygen. Trends Biochem. Sci. 37, 373–380. doi: 10.1016/j.tibs.2012.06.005
Hallberg, B.M., Leitner, C., Haltrich D., Divne, C. (2004) Crystal structure of the 270 kDa homotetrameric lignin-degrading enzyme pyranose 2-oxidase. J. Mol. Biol. 341, 781–796. doi: 10.1016/j.jmb.2004.06.033
Kujawa, M., Ebner, H., Leitner, C., Hallberg, B.M., Prongjit, M., Sucharitakul, J., Ludwig, R., Rudsander, U., Peterbauer, C., Chaiyen, P., Haltrich D., Divne C. (2006) Structural basis for substrate binding and regioselective oxidation of monosaccharides at C3 by pyranose 2-oxidase. J. Biol. Chem. 281, 35104–35115. doi: 10.1074/jbc.M604718200
Massey, V. (1994) Activation of molecular oxygen by flavins and flavoprotein. J. Biol. Chem. 269, 22459–22462. PMID: 8077188
Reetz, M.T., Bocola, M., Carballeira, J.D., Zha, D., Vogel, A. (2005) Expanding the range of substrate acceptance of enzymes: combinatorial active-site saturation test. Angew. Chem. Int. Ed. 44, 4192-4196. doi:10.1002/anie.200500767
