Impact of extrinsic production factors on mammalian cell production performance
SUPERVISOR: RENATE KUNERT
Background.
In the last three decades, cell culture medium optimization was the most successful way of improving the expression yield in mammalian cell cultivation. It was possible to increase volumetric mAb titres hundred fold – even though supplemented fetal calf serum (FCS), providing highly demanded growth factors and hormones, was replaced by small and chemically defined media components without any proteins or hydrolysates thereof. The balance of added lipid precursors, (non-)essential amino acids, vitamins and cofactors as well as trace elements is highly sophisticated and very often commercial media formulations are specifically supporting only individual host cell lines whereas for other hosts different formulations are superior (Reinhart et al., 2015). The reason for limited cellular performance of such low-grade media formulation is most often not analysed in detail probably since the ingredients of regularly used chemically defined media are proprietary and according information not available to the end-customer (i.e. scientist).
Based on all published investigations to relieve bottlenecks in secretion and aggregation of therapeutic proteins the optimization strategies remain trial and error (Zhou et al., 2018), but an increasing number of papers define various medium supplements as critical for the protein quality, not only glycosylation but also protein aggregation, formation of low molecular weight variants or amino acid modification (Brühlmann et al., 2015). In reality, scientific labs are still using FCS-containing culture media for many applications dealing with cell based in vitro assays or small scale protein expression, because chemically defined media are very expensive and such there is no benefit to change the FCS-systems. In contrast, in case of clone development for biopharmaceutical large scale production serum free media are inevitable and must be applied even if the prize is above 50 Euro per litre. Despite numerous approaches of single gene engineering that exist, we believe that a more general approach reflects the complex cellular biology resulting in various growth, productivity and metabolic phenotypes by proper medium supplementation.
Aims and methods.
Our working group has developed numerous recombinant cell lines expressing IgGs, IgMs, IgAs, hGH, Epo, FSH, gp140 (the soluble form of HIV env gp160) or CD19-Fc (Chromikova et al., 2015; Gach et al., 2010; Kunert et al., 2004 and 2009; Reinhart et al., 2015). Early cell lines were developed in FCS-containing media and afterwards adapted to serum free and chemically defined media. Other clones were originally cultivated in soy hydrolysate-containing media and then switched to chemically defined media, but it is always trial and error until one has found the optimal production media and afterwards it is not known which components of the commercial medium are responsible for a specific performance.
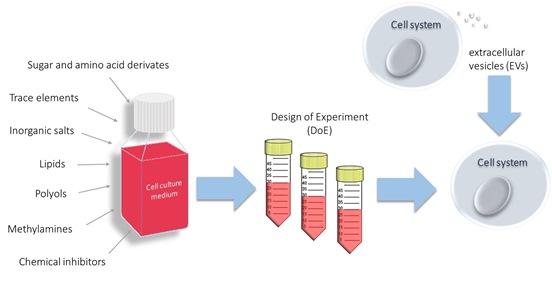
In this thesis we will systematically test different media supplements clustered in different groups like lipids, trace elements, inorganic salts, polyols, sugar and amino acid derivatives, methylamines or chemical inhibitors of cell cycle check points (Du et al., 2015, Johari et al., 2015) and then test in a design-of experiment-approach their suitability in a panel of various clones including difficult to produce and more simple proteins. These substance classes can then be defined for supplementation of the basal media in (spiked) batch experiments, formulated as concentrated feed solutions to be used in high-performing fed-batch cultures using optimal feeding strategies or used as media supplement for perfusion cultures. Combined with novel process analytical tools, including spectral methods such as RAMAN, protein expression and –quality will be investigated in detail related to cellular properties and needs.
Another approach will evaluate the biological potential of so-called conditioned cell culture supernatants. Since decades a simple method to improve the (sub-)cloning efficacy of CHO cells is to supplement the medium with 50% conditioned cell culture supernatant from cells in the exponential growth phase. It is now for the first time published that extracellular vesicles (EVs) inhibit apoptosis in CHO cells (Han et al., 2018) and our goal is to investigate if concentrated fractions of CHO-derived EVs have a higher impact on low density or on high density CHO cultures and how they are interacting with the cell cycle and other regulatory mechanisms.
Collaboration within this thesis involves ALTMANN.
Brühlmann, D., Jordan, M., Hemberger, J., Sauer, M., Stettler, M., Broly, H. (2015) Tailoring recombinant protein quality by rational media design. Biotechnol. Progr. 31, 615-629 doi: 10.1002/btpr.2089
Chromikova, V., Mader, A., Steinfellner, W., Kunert, R. (2015) Evaluating the bottlenecks of recombinant IgM production in mammalian cells. Cytotechnology 67, 343-356. doi:10.1007/ s10616-014-9693-4
Du. Z., Treiber, D., McCarter, J.D., Fomina-Yadlin, D., Saleem, R.A., McCoy, R.E., Zhang, Y., Tharmalingam, T., Leith, M., Follstad, B.D., Dell, B., Grisim, B., Zupke, C., Heath, C., Gach, J.S., Furtmüller, P.G., Quendler, H., Messner, P., Wagner, R., Katinger H., Kunert R. (2010) Proline is not uniquely capable of providing the pivot point for domain swapping in 2G12, a broadly neutralizing antibody against HIV-1. J. Biol. Chem. 285, 1122-1127. doi: 10.1074/jbc.M109.058792
Han, S. and Rhee, W.J. (2018) Inhibition of apoptosis using exosomes in Chinese hamster ovary cell culture. Biotechnol. Bioeng. 115, 1331-1339. doi: 10.1002/bit.26549
Johari, Y.B., Estes, S.D., Alves, C.S., Sinacore, M.S., James, D.C. (2015) Integrated cell and process engineering for improved transient production of a "difficult-to-express" fusion protein by CHO cells. Biotechnol. Bioeng. 112, 2527-2542. doi: 10.1002/bit.25687
Kunert, R., Wolbank, S., Stiegler, G., Weik R., Katinger H. (2004) Characterization of molecular features, antigen-binding, and in vitro properties of IgG and IgM variants of 4E10, an anti-HIV type 1 neutralizing monoclonal antibody. AIDS Res. Human Retrovir. 20, 755-762. doi:10.1089/0889222041524571
Kunert, R., Steinfellner, W., Altmann, F., Wallner, J., Katinger H., Vorauer-Uhl, K. (2009) CHO-recombinant human growth hormone as a protease sensitive reporter protein. Appl. Microbiol. Biotechnol. 84, 693-699. doi: 10.1007/s00253-009-1997-5
Reinhart, D., Damjanovic, L., Kaisermayer, C., Kunert, R. (2015) Benchmarking of commercially available CHO cell culture media for antibody production. Appl. Microbiol. Biotechnol. 99, 4645-4657. doi: 10.1007/s00253-015-6514-4
Reinhart, D., Kunert, R., (2015) Upstream and downstream processing of recombinant IgA. Biotechnol. Lett. 37, 241-251. doi: 10.1007/s10529-014-1686-z
Zhou, Y., Raju, R., Alves, C., Gilbert, A. (2018) Debottlenecking protein secretion and reducing protein aggregation in the cellular host. Curr. Opin. Biotechnol. 29, 151-157. doi: 10.1016/j.copbio.2018.01.007.
