Cytochromes as electron transfer mediators for bioelectrodes
PRINCIPAL INVESTIGATOR: ROLAND LUDWIG
Background.
Since the introduction of the biosensor concept by Leland C. Clark (Clark and Lyons, 1962) numerous enzymes have been tested as biorecognition elements for biosensors, but only a few form the basis of commercially successful products. The use of enzymes in biosensors may be hampered in many ways, with the most important aspect being a good electric connection between the biorecognition element and the transducing electrode (Heller and Feldman, 2008). Several methods have been developed to electrically connect enzymes to electrodes, and the most specific approach is the direct communication of enzyme and electrode via direct electron transfer (DET) (Das et al., 2016).
In recent years, we have intensely investigated cellobiose dehydrogenase as a DET-capable biorecognition element in glucose and lactose sensing (Ortiz et al., 2017; Kracher et al., 2015; Ludwig et al., 2013). This enzyme has a build-in-mediator in form of an N-terminal cytochrome domain (Kracher et al., 2016). In mitochondrial, membrane-bound electron transfer chains, cytochromes (CYT) play an important role to ensure an efficient flow of electrons between donor and acceptor enzymes.
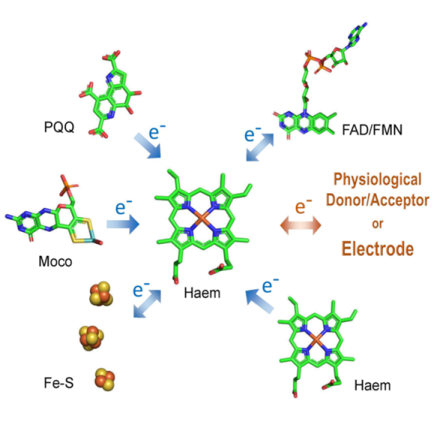
Furthermore, cytochrome c was the first protein for which DET was shown (Eddowes and Hill, 1977) and has become the model cytochrome for electrochemical studies. In the meantime many more cytochromes were described and are accessible by recombinant protein expression.
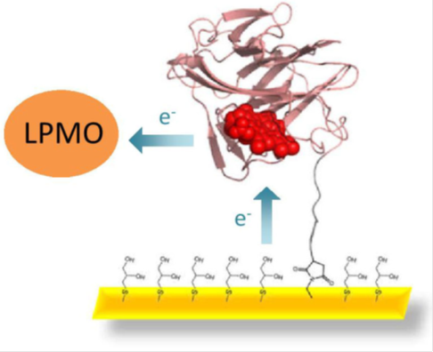
The project will produce bacterial, yeast and fungal cytochromes and investigate the molecular mechanism that enables CYTs to contact oxidoreductases by DET and shuttle the obtained electron to the electrode. Lytic polysaccharide monooxygenase (LPMO) is an important enzyme in cellulose and hemicellulose degradation (Couturier et al., 2018). It is not capable of DET and will be used as a model oxidoreductase in this study. If successful, a route for the production of cellulose and hemicellulose biosensors would be opened. The interaction of suitable CYTs with both an LPMO and the electrode will be investigated for different electrode architectures employing LPMO and CYT in either free or electrode-bound form. Architectures with free LPMO and free/bound CYT will allow the measurement of polymeric substrates, whereas electrode-bound LPMO and CYT will allow only the measurement of soluble carbohydrates. The project aims to elucidate the protein-protein interaction mechanism, electron transfer mechanism, and the mobility of CYT by fast kinetic and electrochemical methods to investigate the potential of CYT to realise LPMO-based biosensors for analytical application in biomass hydrolysis and biofuel production.
Aims and methods.
In this thesis project, we will study the electron transfer between LPMO and different CYTs. After identification of suitable CYTs the efficiency of the DET process to various electrode surfaces will be characterised before assembling model biosensors for cellulose and hemicelluloses. CYTs of bacterial, yeast and fungal origin will be selected from literature and in silico screening of available genomes. Phylogeny and homology models will be used to classify CYTs especially for the expected redox potential, and different CYTs will be selected and expressed in bacterial or yeast expression systems. We have experience with the expression of haemoproteins in various expression hosts (Ma et al., 2017). The modification of CYTs by N- or C-terminal linker peptides for electrode immobilisation will be performed by overlap extension PCR using linker sequences of various lengths. Chromatographic methods for the protein purification are established and can be adapted to the necessary scale of protein production. The interaction mechanism of CYTs and LPMO will be studied by docking and molecular dynamics simulations (OOSTENBRINK). Protein interactions and electron transfer between proteins will be measured in solution by fast kinetic methods (stopped-flow spectroscopy) and thermodynamic methods (ITC) (HALTRICH, OBINGER). The interaction mechanism will be elucidated for the most efficient LPMO/CYT pair by site-directed mutagenesis of amino acid residues at the protein-protein interface.
The electrochemical investigation of the DET from CYT to various electrode surfaces will be based on different potential sweep and amperometric methods using flow-cells, rotating disc electrodes and microelectrodes. To this purpose CYTs will be investigated either in free form or bound onto the electrode. The connection to the electrode will be realised by an N- or C-terminal peptide linker attached to the CYT to allow for a certain mobility and reorientation. Coupling can be performed either by thiol linkage (Matsumura et al., 2012), diazonium coupling (Tasca et al., 2011) or by using maleimide (Al-Lolage et al., 2017). These different coupling methods allow the use of different electrode materials such as gold, pyrolytic graphite, glassy carbon or carbon nanotubes. Different linker lengths and surface modifications (charges and polarity) will be realised to optimise the DET rate between LPMO and CYT as well as CYT and the electrode. Finally, the best sensor architectures will be employed in biomass saccharification studies to evaluate their potential for the detection of substrates and intermediates in large-scale biomass conversion processes.
Collaborations within this thesis will include OOSTENBRINK (molecular dynamics), HALTRICH (fast kinetics) and OBINGER (enzymatic mechanisms).
Al-Lolage, F.A., Meneghello, M., Ma, S., Ludwig, R., Bartlett, P.N. (2017) A flexible method for the stable, covalent immobilization of enzymes at electrode surfaces. Chem. Electro. Chem. 4, 1528-1534. doi: 10.1002/celc.201700135
Clark, L.C. and Lyons, C. (1962) Electrode systems for continuous monitoring in cardiovascular surgery. Ann. N.Y. Acad. Sci. 102, 29-45. doi: 10.1111/j.1749-6632.1962.tb13623.x
Couturier, M., Ladevèze, S., Sulzenbacher, G., Ciano, L., Fanuel, M., … Berrin, J.-G. (2018) Lytic xylan oxidases from wood-decay fungi unlock biomass degradation. Nat. Chem. Biol. 14, 306-310. doi: 10.1038/nchembio.2558
Das, P., Das, M., Chinnadayyala, S.R., Singha, I.M., Goswami, P. (2016) Recent advances on developing 3rd generation enzyme electrode for biosensor applications. Biosens. Bioelectron. 79, 386-397. doi: 10.1016/j.bios.2015.12.055
Eddowes, M.J., Hill, H.A.O. (1977) Novel method for the investigation of the electrochemistry of metalloproteins: cytochrome c. J. Chem. Soc. Chem. Commun. 21, 771b-772. doi: 10.1039/C3977000771b
Heller, A., Feldman, B. (2008) Electrochemical glucose sensors and their applications in diabetes management. Chem. Rev. 108, 2482-2505. doi: 10.1021/cr068069y
Kracher, D., Zahma, K., Schulz, C., Sygmund, C., Gorton, L., Ludwig, R. (2015) Inter-domain electron transfer in cellobiose dehydrogenase: modulation by pH and divalent cations. FEBS J. 282, 3136-3148. doi: 10.1111/febs.13310
Kracher, D., Scheiblbrandner, S., Felice, A.K.G., Breslmayr, E., Preims, M., Ludwicka, K., Haltrich, D., Eijsink, V.G.H., Ludwig, R. (2016) Extracellular electron transfer systems fuel cellulose oxidative degradation. Science, 352, 1098-1101. doi: 10.1126/ science.aaf3165
Ludwig, R., Ortiz, R., Schulz, C., Harreither, W., Sygmund, C., Gorton, L. (2013) Cellobiose dehydrogenase modified electrodes: advances by materials science and biochemical engineering. Anal. Bioanal. Chem. 405, 3637-3658. doi: 10.1007/s00216-012-6627-x
Ma, S., Preims, M., Piumi, F., Kappel, L., Seiboth, B., Record, E., Kracher, D., Ludwig, R. (2017) Molecular and catalytic properties of fungal extracellular cellobiose dehydrogenase produced in prokaryotic and eukaryotic expression systems. Microb. Cell Factor. 16, 37. doi: 10.1186/s12934-017-0653-5
Matsumura, H., Ortiz, R., Ludwig, R., Igarashi, K., Samejima, M., Gorton, L. (2012) Direct electrochemistry of Phanerochaete chrysosporium cellobiose dehydrogenase covalently attached onto gold nanoparticle modified solid gold electrodes. Langmuir, 28, 10925-10933. doi: 10.1021/la3018858
Ortiz, R., Rahman, M., Zangrilli, B., Sygmund, C., Micheelsen, P.O., Silow, M., Toscano, M.D., Ludwig, R., Gorton, L. (2017) Engineering of class II cellobiose dehydrogenases for improved glucose sensitivity and reduced maltose affinity. Chem. Electro. Chem. 4, 750. doi: 10.1002/celc.201700226
Tasca, F., Harreither, W., Ludwig, R., Gooding, J.J., Gorton, L. (2011) Cellobiose dehydrogenase aryl diazonium modified single walled carbon nanotubes: enhanced direct electron transfer through a positively charged surface. Anal. Chem. 83, 3042-3049. doi: 10.1021/ac103250b
