Routing the flow of electron from cytochromes to oxidoreductases
PRINCIPAL INVESTIGATOR: Roland LUDWIG
Background.
Cytochromes are small, heme carrying redox proteins, which make them the main proponents of the electron transfer between enzymes. In their function as electron transfer mediators, cytochromes fulfill key functions in photosynthetic and other metabolic processes. Because of their function cytochromes are frequently anchored in membranes of chloroplasts or mitochondria, but they are also found in the cytoplasm. Cytochromes can interact with PQQ, FAD, FMN, heme, Moco, and FeS carrying enzymes to transfer electrons (Ma and Ludwig 2019).
Oxidoreductases are a major enzyme class targeted in biosensor development, because their substrates are physiologically and industrially relevant and they can be coupled to electrodes via mediated electron transfer (MET) or direct electron transfer (DET) (Zayats et al. 2008, Willner et al. 2009). The electron transfer efficiency, mass transfer and applied overpotential determine the generated signal current, which defines the biosensor’s sensitivity and limit of detection (Turner, A.P.F. 2013). Efficient coupling of oxidoreductases is typically achieved at a potential difference of 50 – 100 mV between the enzymes redox cofactor and the electrode (Lindgren et al. 2000). If the potential difference is to high the biosensor becomes more sensitive towards electroactive, interfering matrix compounds.
For the glucose biosensor several enzymes have been employed as electroactive biocomponent, but especially glucose oxidase (GOX) and glucose dehydrogenase (GDH) from the GMC-oxidoreductase superfamily (Cavener, D.R. 1992). Numerous architectures for second- (MET), and third-generation (DET) glucose biosensors have been proposed and discussed (Wang, J. 2005). The critical factor is the electrical coupling of the oxidoreductase activity to the transducing electrode. Free or immobilized redox mediators can introduce several problems of their own, e.g. diffusion limitation, stability, poisoning by matrix compounds, unfitting redox potentials, etc (Heller, A. 2006). Enzymes that are capable of DET are independent of redox mediators. The aim of this thesis project is to investigate the electron transferring properties of recombinant or engineered cytochromes (CYT) and to fuse them to glucose oxidase and glucose dehydrogenase. To achieve efficient interdomain electron transfer (IET) between the catalytic subunit and the electron transferring cytochrome subunit, their interaction will be studied and optimized by rational engineering. Finally, optimized biorecognition elements will be employed in various biosensor architectures to improve DET to the electrode and to produce and preliminary characterize the glucose biosensors based on the fusion enzymes.
Aims and methods.
In this thesis project, the electron transfer of different cytochromes (CYT) and the glucose converting GMC-oxidoreductases glucose oxidase (from Aspergillus niger) and glucose dehydrogenase (from Glomerella cingulata) will be studied. First, suitable CYTs will be selected by structural and electrostatic complementarity to the GMC-oxidoreductase interface around the substrate channel. Expression of potentially applicable CYT’s will be performed in bacterial or yeast expression systems. We have experience with the expression of haemoproteins in various expression hosts (Ma et al. 2017). Chromatographic methods for the protein purification are established and can be adapted to the necessary scale of protein production.
The DET efficiency of purified CYTs to gold and carbon electrode surfaces will be characterized before fusing the CYTs to GOX or GDH by potential step and potential sweep methods using flow-cells and rotating disc electrodes. Only CYTs with a well-fitting redox potential and fast DET will be selected to generate CYT-GMC-oxidoreducatase fusion enzymes. The interaction mechanism of CYTs and the GMC-oxidoreductases will be studied by docking and molecular dynamics simulations (collaborator: OOSTENBRINK). Only cytochromes showing a good sterical- and electrostatic complementarity to the enzyme interaction-site will be selected for the generation of fusion enzymes. The fusion enzymes generated in the first round will be characterized in regard to their domain interaction and electron transfer in solution by fast kinetic methods (stopped-flow spectroscopy) and thermodynamic methods (ITC) (collaborators: HALTRICH, OBINGER).
The most efficient CYT-GMC-oxidoreductases pair in regard to IET will form the basis for the second round of optimization. To increase IET, the optimal length of the protein linker between both domains will be elucidated. Similarly, the steric- and electrostatic complementarity of the two domains will be fine-tuned by amino acid exchange. The optimized CYT-GMC-oxidoreductase fusion enzyme will be fully characterized in regard to its catalytic and electron transfer mechanism by the above mentioned kinetic and electrochemical methods. It will also be immobilized on several gold and carbon electrode surfaces to determine its DET, but also its stability, selectivity, and sensitivity as a biorecognition element. Connection to the electrode will be realised by either unspecific adsorbtion, encapsulation in a polymer or covalent coupling via a thiol linkage (Matsumura et al. 2012), diazonium coupling (Tasca et al. 2011) or by using maleimide (Al-Lolage et al. 2017). Finally, the best sensor architecture will be characterized and compared to existing glucose biosensors.
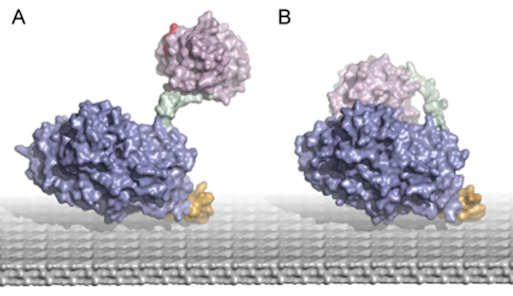
Figure. A schematic view of a mobile cytochrome domain (pink) which is attached to the cytalytic GMC-oxidoreductase domain (violet) via a flexible linker (green). The domain movement between open conformation (A) and closed conformation (B) is indicated.
Collaborations within this thesis include OOSTENBRINK, HALTRICH and OBINGER
Al-Lolage, F.A., Meneghello, M., Ma, S., Ludwig, R., Bartlett, P.N. (2017) A flexible method for the stable, covalent immobilization of enzymes at electrode surfaces. Chem. Electro. Chem. 4, 1528 - 1534. doi: 10.1002/celc.201700135
Cavener, D.R. (1992) GMC oxidoreductases. A newly defined family of homologous proteins with diverse catalytic activities. J. Mol. Biol. 223:811-4. doi: 10.1016/0022-2836(92)90992-S
Heller, A. (2006) Electron-conducting redox hydrogels: design, characteristics and synthesis. Curr. Opin. Chem. Biol. 10:664-72. Doi: 10.1016/j.cbpa.2006.09.018
Lindgren, A., Larsson, T., Ruzgas, T., Gorton, L. (2000) Direct electron transfer between the heme of cellobiose dehydrogenase and thiol modified gold electrodes. J Electroanal. Chem. 494:105-113. doi: 10.1016/S0022-0728(00)00326-0
Ma, S., Ludwig, R. (2019) Direct electron transfer of enzymes facilitated by cytochromes. Chem. Electro. Chem. 6: 1-19. doi: 10.1002/celc.201801256
Ma, S., Preims, M., Piumi, F., Kappel, L., Seiboth, B., Record, E., Kracher, D., Ludwig, R. (2017) Molecular and catalytic properties of fungal extracellular cellobiose dehydrogenase produced in prokaryotic and eukaryotic expression systems. Microb. Cell Factor. 16, 37, doi: 10.1186/s12934-017-0653-5
Matsumura, H., Ortiz, R., Ludwig, R., Igarashi, K., Samejima, M., Gorton, L. (2012)
Direct electrochemistry of Phanerochaete chrysosporium cellobiose dehydrogenase covalently attached onto gold nanoparticle modified solid gold electrodes. Langmuir, 28, 10925 - 10933. doi: 10.1021/la3018858
Tasca, F., Harreither, W., Ludwig, R., Gooding, J.J., Gorton, L. (2011) Cellobiose dehydrogenase aryl diazonium modified single walled carbon nanotubes: enhanced direct electron transfer through a positively charged surface. Anal. Chem. 83, 3042 - 3049. doi: 10.1021/ac103250b
Turner, A.P.F. (2013) Biosensors: sense and sensibility. Chem Soc Rev. 42:3184-3196. doi: 10.1039/c3cs35528d
Willner, I., Yan, Y.M., Willner, B., Tel-Vered R. (2009) Integrated enzyme-based biofuel cells - a review. Fuel Cells, 9:7-24. doi: 10.1002/fuce.200800115
Zayats, M., Willner, B., Willner, I. (2008) Design of amperometric biosensors and biofuel cells by the reconstitution of electrically contacted enzyme electrodes. Electroanal. 20:583-601. doi:10.1002/Bit.23041
