Unbreakable - introducing different enzymatic functions into a highly stable heme-binding protein from Sulfolobus solfataricus
PRINCIPAL INVESTIGATOR: CHRISTIAN OBINGER
Background.
Sulfolobus solfataricus is a thermoacidophilic Archeon that grows between 55 and 90 °C, at a pH optimum between 2 and 3 and requires an aerobic environment (Brock et al., 1972). Consequently, proteins expressed by this organism are hyperthermostable. SsoHBP (heme binding protein from S. solfataricus) can further tolerate highly acidic conditions, even though the cytosolic pH in S. solfataricus is maintained at 6.5. The Tm value at pH 2.5 for SsoHBP is at 109°C and the iron-protein does not form any disulphide bonds.
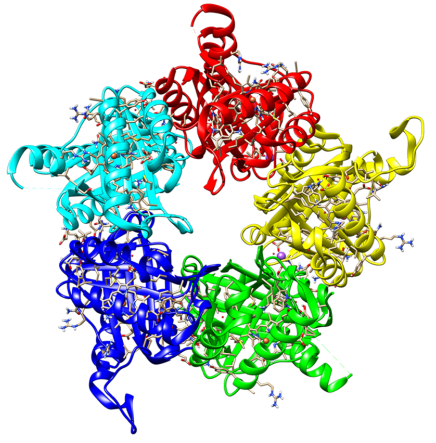
SsoHBP belongs to the α,β-barrel-domain structural superfamily and shares a common ferredoxin-like fold with chlorite dismutases (Clds), coproheme decarboxylases (ChdCs) and dye-decolorizing peroxidases (DyPs) (Hofbauer et al., 2014; Celis and DuBois, 2015). The overall structure within this structural superfamily is highly conserved, with the exception of a flexible stretch that forms the active site access channel. SsoHBP was shown to bind heme and coproheme to form high spin complexes, but did not exhibit any significant Cld activity (ClO2- → O2 + Cl-), DyP activity (H2O2 + 2 AH → 2 H2O + 2 A•) nor ChdC activity (coproheme → heme b) (Pfanzagl et al., 2018). Its physiological role is completely unknown.
Aims and methods.
This project aims at (i) characterization and identification of the yet unknown enzymatic function(s) of SsoHBP, (ii) understanding the structural basis of the high conformational and thermal stability of SsoHBP, and at (iii) the introduction of different enzymatic functions by rationally engineering the proximal and distal heme environment and the above mentioned loop that forms the substrate channel. The project takes advantage of SsoHBP’s thermostability and pH-tolerance and the expertise of the OBINGER group in structure-function relationships of ChdCs, Clds and DyPs.
Proteins from S. solfataricus have proven to be perfect, hyperstable scaffolds for various protein engineering approaches (Traxlmayr et al., 2016; Vettone et al., 2016). Based on our knowledge about structure and function of Clds, ChdCs and DyPs we aim at engineering the heme cavity of SsoHBP to introduce the respective activities. For example we aim at introducing chlorite dismutase activity by exchanging the distal residue of SsoHBP (Ser → Arg) and rebuilding the Cld-typical H-bonding network on the proximal heme side. Additionally, we shall insert a distal base for deprotonation of hydrogen peroxide (e.g. Asp as in DyPs) as a basis for its heterolytic cleavage etc. Modified SsoHBP scaffolds will deliver starting points for further protein engineering approaches yielding enzymes being suitable for industrial applications at extreme conditions [bioremediation of waste water (e.g. chlorite degradation), environmental friendly bleaching of dyes, etc.].
SsoHBP (wild-type and variants) will be heterologously expressed in E. coli. Based on available X-ray structures from Clds, ChdCs and also dye-decolorizing peroxidases (DyPs), site-directed mutagenesis will be performed. Emphasis will be on amino acids at positions of catalytic residues of related enzyme families, as well as on the alteration of the substrate channel forming flexible loop. Electron migration pathways will be introduced in silico to facilitate long range electron transfer from the heme to the protein surface in cooperation with Chris OOSTENBRINK and later realized in the lab.
Wild-type and mutant proteins will be characterized by a broad set of biochemical/physical methods including (i) enzymatic activity assays for different catalytic functions at various conditions, (ii) structural investigations by X-ray crystallography (in close cooperation with Kristina DJINOVIC-CARUGO from the Department for Structural and Computational Biology, Max F. Perutz Laboratories, University of Vienna), (iii) detailed spectral analysis (UV-Vis, electron paramagnetic resonance and resonance Raman spectroscopy) of proteins, and protein-based radicals in different redox- and spin-states (in cooperation with Sabine VAN DOORSLAER and with Giulietta SMULEVICH), (iv) time-resolved multi-mixing UV-Vis stopped-flow studies in order to analyse the kinetics of interconversion and spectroscopic features of relevant redox intermediates, (v) spectroelectrochemical studies (in cooperation with Gianantonio BATTISTUZZI, and Roland LUDWIG from BOKU) and (vi) calorimetric studies using ITC and DSC (available at the BOKU Core Facility Biomolecular and Cellular Analysis).
These complementary methods will provide the basis for understanding of structure-function relationships of SsoHBP and will show its potential to be a versatile scaffold for the introduction of different enzymatic functionalities. Wild-type SsoHBP and variants will be analyzed by classical and ab initio molecular dynamics simulations (in cooperation with OOSTENBRINK).
Collaborations within this thesis will include LUDWIG (electrochemistry) and OOSTENBRINK (protein modelling). National and international collaborations will include DJINOVIC-CARUGO (X-ray crystallography), SMULEVICH (RR spectroscopy), BATTISTUZZI (spectroelectrochemistry) and VAN DOORSLAER (electron paramagnetic spectroscopy).
Brock, T.D., Brock, K.M., Belly, R.T., Weiss, R.L. (1972) Sulfolobus: a new genus of sulfur-oxidizing bacteria living at low pH and high temperature. Arch. Microbiol. 84, 54-68. doi: 10.1007/BF00408082
Celis, A.I. and DuBois, J.L. (2015) Substrate, product, and cofactor: The extraordinarily flexible relationship between the CDE superfamily and heme. Arch. Biochem. Biophys. 574, 3-17. doi: 10.1016/j.abb.2015.03.004
Hofbauer, S., Schaffner, I., Furtmüller, P.G., Obinger, C. (2014) Chlorite dismutases - a heme enzyme family for use in bioremediation and generation of molecular oxygen. Biotechnol. J. 9, 461-473. doi: 10.1002/biot.201300210
Pfanzagl, V., Holcik, L., Maresch, D., Gorgone, G., Michlits, H., Furtmüller, P.G., Hofbauer, S. (2018) Coproheme decarboxylases - Phylogenetic prediction versus biochemical experiments. Arch. Biochem. Biophys. 640, 27-36. doi: 10.1016/ j.abb.2018.01.005
Traxlmayr, M.W., Kiefer, J.D., Srinivas, R.R., Lobner, E., Tisdale, A.W., Mehta, N.K., Yang, N.J., Tidor, B., Wittrup, K.D. (2016) Strong Enrichment of Aromatic Residues in Binding Sites from a Charge-neutralized Hyperthermostable Sso7d Scaffold Library. J. Biol. Chem. 291, 22496-22508. doi: 10.1074/jbc.M116.741314
Vettone, A., Serpe, M., Hidalgo, A., Berenguer, J., del Monaco, G., Valenti, A., Rossi, M., Ciaramella, M., Perugino, G. (2016) A novel thermostable protein-tag: optimization of the Sulfolobus solfataricus DNA- alkyl-transferase by protein engineering. Extremophiles 20, 1-13. doi: 10.1007/s00792-015-0791-9
