In planta engineering and functional activities of oligo-sialic acid
SUPERVISOR: HERTA STEINKELLNER
Background.
Polysialic acid (polySia) is a linear homo-polymer of the sugar sialic acid (Sia) that is omnipresent in eukaryotes. In nature, the degree of polymerization varies significantly reaching levels of up to 400 sialic acid residues. Various studies delivered impressive evidence for the multiple functions of the sugar polymer (free or protein-bound) in immunological and neurological settings (e.g. Werneburg et al., 2017). Importantly, current results point to the impact of the chain length to functional activities, for example polymers with a restricted size (approx. 20 residues) prevent inflammation processes more efficiently than large polymers (Karlstetter et al., 2017). However due to its complexity the controlled synthesis of polysialic acid structures is a challenge for which reliable solutions have not yet been identified.
It is well known that sialyltransferases of the mammalian ST8Sia family catalyse the transfer of sialic acid polymers to glycoproteins. One member (i.e. the oligosyltransferase ST8Sia-III) is known to transfer sialic residues >30 residues (Angata et al., 2000). ST8Sia-III is a Golgi-resident enzyme and the sole prerequisite for building sialic acid chains on their glycoprotein acceptors is the presence of appropriate core N-glycans terminating with α2,3- or 2,6-linked sialic acids (Angata et al., 2000). In recent studies we have shown by the overexpression of human polysialyltransferases ST8Sia-II and IV (members of the ST8Sia family) the synthesis of polySia structures up to 40 residues in plants. Plant-produced polySia is highly active as shown in immunological settings (Kallolimath et al., 2016). We hypothesize that by overexpression of ST8Sia-III the generation of polySia with restricted size (>30 residues) is possible in planta.
Aims and methods.
Here we aim to engineer plants for the synthesis of sialic acid structures with a restricted size (>30 residues) and test their functional activities. We focus on the in planta expression of the (oligo)-sialyltransferase ST8Sia-III, and monitor its ability to elongate protein-bound sialylated N-glycans.
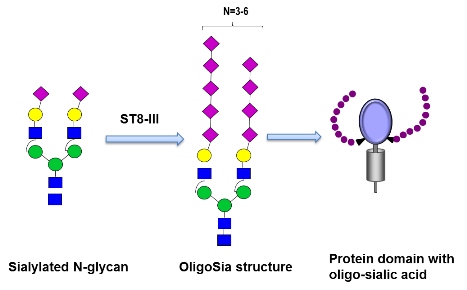
A glyco-engineered plant line (Nicotiana benthamiana-Sia) previously generated in our laboratory that synthesizes the appropriate acceptor substrate (i.e. protein-bound α2,6-, α2,3-linked sialylated complex N-glycans) will be used as expression host (Kallolimath et al., 2016), and human alpha-2-HS-glycoprotein (also known as fetuin) will be used as target protein for the transfer of sugar oligo/polymers. This protein, which was previously used for in vitro monitoring of ST8III activities, seems to be an appropriate acceptor and has the advantage that it contains N-glycans and O-glycans (Angata et al., 2000). Although in this proposal the main target is N-glycan sialylation, fetuin also offers the possibility to determine O-glycan oligo/polysialylation, an engineering approach applied recently by the PI (Castilho et al., 2012). The functional impact of oligo/polymers will be evaluated in appropriate immunological settings (impact of sialylation in receptor-specific inflammatory activation) and compared to activities induced by polySia compounds of <40 residues, generated by the overexpression of ST8Sia-II and IV. This allows a side-by-side comparison of sialic acid chains with different chain lengths.
cDNAs of ST8Sia-III and fetuin will be cloned into appropriate vectors and transiently expressed in the glycosylation mutant N. benthamiana-Sia. Various settings will be monitored to obtain efficient protein sialylation, e.g. co-expression of oligosaccharyltransferase, an enzyme that increases the recognition of protein N-glycosylation sites (Castilho et al., 2018). Also the impact of targeting of ST8Sia-II and IV to different sub Golgi compartments will be monitored as the precise subcellular localisation of glycosyltransferases has significant impact on the targeted formation of glycan structures. The synthesis of sialic acid oligo and polymers will be determined by immunological means using appropriate antibodies. Tag-affinity purified fetuin will be used for precise determination of size, structure, and chemical nature of sialylated compounds. Methods employed will include fluorescent HPLC, MALLS, NMR (in cooperation with Altmann). A biochemical and biophysical characterization of sialylated fetuin (e.g. oligomerisation status, thermal stability, Obinger) will be carried out to reveal possible glycan-associated modifications. Functional activity studies (in cooperation with Gerardy Schahn and Herbert Hildebrandt, Medical School Hannover, GER) include the assessment of anti-inflammatory effects (quantified by NO, TNFα and IL-1ß production) in cellular models (Kallolimath et al. 2016). Experimental data will be evaluated in the context of Computer-based simulation and modelling with the aim to rationalise experimental data (in cooperation with Oostenbrink).
Collaborations in this thesis involve ALTMANN (glycoanalytics), OOSTENBRINK (glycan modelling and simulation) and OBINGER (structural/functional analysis of proteins).
Angata, K., Suzuki, M., McAuliffe, J., Ding, Y., Hindsgaul, O., Fukuda, M. (2000) Differential biosynthesis of polysialic acid on NCAM and oligosaccharide acceptors by α2,8-sialyltransferases, ST8Sia IV, ST8Sia II, and ST8Sia III. J. Biol. Chem. 275, 18594-18601. doi: 10.1074/jbc.M910204199
Castilho, A., Beihammer, G., Pfeiffer, C., Göritzer, K., Montero-Morales, L., Maresch, D., Grünwald-Gruber, C., Altmann, F., Steinkellner, H., Strasser, R. (2018) An oligosaccharyltransferase from Leishmania major increases N-glycan occupancy. Plant Biotechnol. J., doi: 10.1111/pbi.12906
Castilho, A., Neumann, L., Daskalova, S., Mason, H.S., Steinkellner, H., Altmann, F., Strasser, R. (2012) Engineering of sialylated mucin-type O-glycosylation in plants. J. Biol. Chem. 287, 36518-36526. doi: 10.1074/jbc.M112.402685
Kallolimath, S., Castilho, A., Strasser, R., Grunwald-Gruber, C., Altmann, F., Strubl, S., Galuska, C.E., Galuska, S.P., Werner, S., Werneburg, S., Hildebrandt, H., Gerardy-Schahn, R., Steinkellner, H. (2016) Engineering of complex protein sialylation in plants. Proc. Natl. Acad. Sci. USA 113, 9498-9503. doi: 10.1073/pnas.1604371113
Karlstetter, M., Kopatz, J., Shahraz, A., Caramoy, A., Lin, Y., Lückoff, A., Fauser, S., Düker, K., Claude, J., Wang, Y., Ackermann, J., Schmidt, T., Hornung, V., Skerka, C., Langmann, T., Neumann, H. (2017) Polysialic acid blocks mononuclear phagocyte reactivity, inhibits complement activation, and protects from vascular damage in the retina. EMBO Mol. Med. 9, 154-166. doi: 10.15252/emmm.201606627
Werneburg, S., Fuchs, H.L.S., Albers, I., Burkhardt, H., Gudi, V., Skripuletz, T., Stangel, M., Gerardy-Schahn, R., Hildebrandt, H. (2017). Polysialylation at Early Stages of Oligodendrocyte Differentiation Promotes Myelin Repair. J. Neurosci. 37, 8131-8141. doi: 10.1523/JNEUROSCI.1147-17.2017
