Engineering and functional activities of polysialylated IgG antibodies
SUPERVISOR: HERTA STEINKELLNER
Background.
Monoclonal antibodies are a success story in translational medicine. This refers particularly to antibodies of the IgG isotype. While a significant number of monoclonal IgGs are being applied in various therapeutic settings a limitation in their broad use is their inability to cross the brain blood barrier (BBB).
However, many neuro-biological settings (e.g. age related neuro-degenerative diseases like Alzheimer disease) would benefit from efficient antibody-based therapies (Honig et al., 2018). Note, the presence of certain sugar polymers (i.e. α2,8-linked polysialic acid, polySia) of neuroinvasive bacteria allows these organisms to evade the host immune system and cross the BBB. This very quality that makes polySia poor in vaccine development is a benefit for its use in the modification of therapeutic proteins. Indeed, polySia−coupled drugs exhibit an increased penetrability to BBB resulting in more potent therapeutic effects (Wang et al., 2016). Moreover, proteins coupled with this sugar polymer exhibit enhanced serum half live due to special physicochemical properties and reduced immunogenicity (Ilyushin et al., 2013).
Here we hypothesize that IgG antibodies that carry a modified glycosylation pattern (i.e. poly-sialylated N-glycans) exhibit beneficial or even novel therapeutic features, like the ability to cross the BBB and increased serum half live.
Aims and methods.
Here we aim to express monoclonal IgG antibodies (mIgGs) that carry α2,8-polysialylated N-glycans attached to their single Fc-located N-glycosylation site.
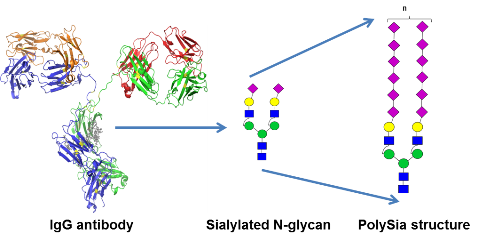
We aim to test the impact of polySia to IgG`s biochemical properties (e.g. thermal stability, assembly) and functional activities, with main emphasis on the ability of IgGs to cross the BBB and to extend the serum half live.
We build on our recent results where we show in planta synthesis of α2,8-polySia by the overexpression of human polysialyltransferases ST8II and IV (Kallolimath et al., 2016). Preliminary studies have shown that ST8II and IV are able to polysialylate proteins other than neural cell adhesion molecule, a native carrier in the human brain (Kallolimath, doctoral dissertation, 2018, non-public results) in planta. Also, we succeeded in the in planta expression of mIgGs with α2,6- α2,3-linked sialylated N-glycans, the appropriate acceptor substrate for sialic acid polymer formation (Castilho et al., 2015). These results serve as basis for the transient expression of mIgGs (and Fc fragments) in glyco-engineered plants in combination with ST8II and IV. Various settings will be monitored to obtain efficient polySia-mIgGs, e.g. co-expression of oligosaccharyltransferase, an enzyme that increases the recognition of N-glycosylation sites (Castilho et al., 2018). Also the impact of targeting ST8II and IV to different sub Golgi compartments will be monitored as the precise subcellular localisation of glycosyltransferases has significant impact on the targeted formation of glycan structures (Castilho et al., 2015).
mIgGs will be affinity purified and subjected to various methods that allow a precise determination of size and structure of polySia (fluorescent HPLC, MALLS, NMR and immunological techniques). Also, purification of polySia using standard procedures (combination of detergent/alcohol/salt precipitations, carbon filtration and chromatographic separations) will be used. A biochemical and biophysical characterization of purified mIgGs (e.g. oligomerisation status, thermal stability) will be carried out to reveal possible glycan-associated modifications of the protein. Pharmacokinetic features and brain penetration of mIgGs will be evaluated in animal experiments. IgGs with and without polySia will be administered to mice by different routes, ELISA and HPLC-based methods are used to determine IgG concentrations in brain, liquor and blood. Animal studies will be performed in cooperation with Rita Gerardy Schahn and Herbert Hildebrandt (Medizinische Hochschue Hannover), Alexander Dityatev (German Center for Neurodegenerative Diseases, GER) and Falk Nimmerjahn (Universität Erlangen-Nürnberg, GER).
Collaborations in this thesis involve ALTMANN (glycoanalytics), RÜKER (antibody engineering), OOSTENBRINK (glycan modelling and simulation) and OBINGER (structural/functional analysis of proteins)
Castilho, A., Beihammer, G., Pfeiffer, C., Göritzer, K., Montero-Morales, L., Maresch, D., Grünwald-Gruber, C., Altmann, F., Steinkellner, H., Strasser, R. (2018) An oligosaccharyltransferase from Leishmania major increases N-glycan occupancy. Plant Biotechnol. J., doi: 10.1111/pbi.12906
Castilho, A., Gruber, C., Thader, A., Oostenbrink, C., Pechlaner, M., Steinkellner, H., Altmann, F. (2015) Processing of complex N-glycans in IgG Fc-region is affected by core fucosylation. mAbs. 7, 863-870. doi: 10.1080/19420862.2015.1053683
Honig, L.S, Vellas. B., Woodward, M. et al. (2018) Trial of Solanezumab for Mild Dementia Due to Alzheimer's Disease. N. Engl. J. Med. 378, 321-330. doi: 10.1056/ NEJMoa1705971
Ilyushin, D.G., Smirnov, I.V., Belogurov, A.A. Jr, et al. (2013). Chemical polysialylation of human recombinant butyrylcholinesterase delivers a long-acting bioscavenger for nerve agents in vivo. Proc. Natl. Acad. Sci. USA 110, 1243-1248. doi: 10.1073/ pnas.1211118110
Kallolimath, S., Castilho, A., Strasser, R., Grunwald-Gruber, C., Altmann, F., Strubl, S., Galuska, C.E., Galuska, S.P., Werner, S., Werneburg, S., Hildebrandt, H., Gerardy-Schahn, R., Steinkellner, H. (2016) Engineering of complex protein sialylation in plants. Proc. Natl. Acad. Sci. USA 113, 9498-9503. doi: 10.1073/pnas.1604371113
Wang, X.J., Gao, Y.P., Lu, N.N., Li, W.S., Xu, J.F., Ying, X.Y., Wu, G., Liao, M.H., Tan, C., Shao, L.X., Lu, Y.M., Zhang, C., Fukunaga, K., Han, F., Du, Y.Z. (2016) Endogenous Polysialic Acid Based Micelles for Calmodulin Antagonist Delivery against Vascular Dementia. ACS Appl. Mater. Interfaces 8, 35045-35058. doi: 10.1021/acsami.6b13052
