Designed protein storage organelles and their uptake into target cells
SUPERVISOR: EVA STÖGER
Background.
Biopolymer-based nanoparticles are widely used as carrier for the delivery of therapeutic molecules. A variety of materials and preparation methods have been developed for application-specific properties in terms of particle shape, surface charge, and surface features.
Among the protein-based polymers, zein isolated from maize seeds has been widely used as a carrier because of favorable properties such as biocompatibility, insolubility and low water uptake, mechanical and chemical stability, and its propensity to form coatings and microparticles (Liu et al., 2005). Zein is also generally recognised as safe (GRAS) for food use and resists digestion, making it particularly suitable as an encapsulation polymer for oral drugs (Ahmed et al., 2015). The intravenous delivery of drug-loaded zein-based microparticles has also been investigated as a means to achieve long-acting effects such as the slow and sustained release of pharmaceutical compounds and more efficient drug delivery to cancer cells (Lohcharoenkal et al., 2014). Zein-based microspheres may also provide adjuvant effects when used as vaccine carriers (Hurtado-Lopez and Murdan, 2006; Hofbauer et al., 2016). An adjuvant effect conferred by a polymer-forming protein domain has also been described, and particulate antigens are transported more efficiently to murine splenic follicular dendritic cells in vivo, making them more immunogenic than soluble antigens (Link et al., 2012). The in vitro loading of zein-based microparticles with drugs usually involves spray or freeze drying or liquid–liquid dispersion methods. These technical processes are expensive and can affect the activity of the encapsulated agent, e.g., the high temperatures required for spray drying are incompatible with many pharmaceutical proteins. It is therefore appealing to use plants to achieve microencapsulation in vivo by directly incorporating recombinant proteins into protein storage organelles (Hofbauer and Stoger, 2013).
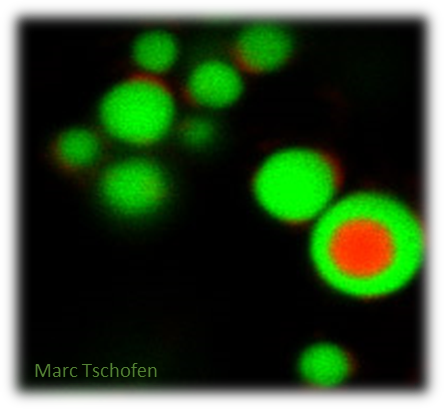
The induction of ectopic protein bodies in leaf tissues as a consequence of aggregation or overexpression of recombinant proteins suggests that PB formation can be readily induced in a rather unspecific process, and this has been exploited with various pharmaceutical proteins (Hofbauer et al., 2016; Saberianfar et al., 2017). However, the design of repurposed multi-component PBs with a defined structure has not been widely explored yet, and the mechanism and efficiency of uptake into target cells remains to be further studied.
Aims and methods.
Based on the understanding of the factors essential for efficient PB formation we will design ectopic PBs consisting of several protein layers with different resistance to degradation and digestion, and we will include recombinant pharmaceutical proteins in order to achieve bioencapsulation and controlled release upon administration. For this we will use model vaccines and therapeutic enzymes for oral application, together with different combinations of seed storage proteins for encapsulation.
A special focus will be placed on candidate vaccines for veterinary applications (Kolotilin et al., 2014). The type, ratio and temporal expression pattern of the proteins used for encapsulation will be varied to achieve different release kinetics. The structure and morphology of the resulting PBs will be investigated by confocal as well as 2D and 3D electron microscopy. Spectroscopic and calorimetric techniques will be used to obtain structural information for the recombinant proteins, and this will be done in collaboration with OBINGER. Uptake into target cells for oral or nasal application will be studied using human intestinal epithelial cell lines and human bronchial epithelial cell lines, both available in the lab of GRILLARI. The mechanism of uptake will be determined by confocal microscopy using fluorescent subcellular labels and dyes suitable for live cell imaging. Phagocytosis of PB-encapsulated antigens by macrophages and dendritic cells will be assessed. Animal intestinal models and biopsies will be used to investigate the uptake and processing of recombinant PBs for specific applications. This will be done together with international collaboration partners in Norway.
Outcome: Repurposed multi-component protein bodies with defined structures and uptake kinetics for the bioencapsulation of pharmaceutical proteins.
Collaborations within this thesis will include GRILLARI (mammalian cell culture models), OBINGER (spectroscopic and calorimetric techniques), GRABHERR (immunology and epitope presentation) and BORTH (Fluorescence-activated cell sorting).
Ahmed, O.A., Hosny, K.M., Al-Sawahli, M.M., Fahmy, U.A. (2015) Optimization of caseinate-coated simvastatin-zein nanoparticles: improved bioavailability and modified release characteristics. Drug. Des. Devel. Ther. 9, 655-662. doi: 10.2147/DDDT.S76194
Hofbauer, A., Melnik, S., Tschofen, M., Arcalis, E., Phan, H.T., Gresch, U., Lampel, J., Conrad, U., Stoger, E. (2016) The Encapsulation of Hemagglutinin in Protein Bodies Achieves a Stronger Immune Response in Mice than the Soluble Antigen. Front. Plant Sci. 7, 142. doi: 10.3389/fpls.2016.00142
Hofbauer, A. and Stoger, E. (2013) Subcellular Accumulation and Modification of Pharmaceutical Proteins in Different Plant Tissues. Curr. Pharm. Des. 19, 5495-5502. doi: 10.2174/1381612811319310005
Hurtado-Lopez, P. and Murdan, S. (2006) Zein microspheres as drug/antigen carriers: a study of their degradation and erosion, in the presence and absence of enzymes. J. Microencaps. 23, 303-314. doi:10.1080/02652040500444149
Kolotilin, I., Topp, E., Cox, E., Devriendt, B., Conrad, U., Joensuu, J., Stoger, E., Warzecha, H., McAllister, T., Potter, A., McLean, M.D., Hall, J.C., Menassa, R. (2014) Plant-based solutions for veterinary immunotherapeutics and prophylactics. Vet. Res. 45, 117. doi: 10.1186/s13567-014-0117-4
Link, A., Zabel, F., Schnetzler, Y., Titz, A., Brombacher, F., Bachmann, M.F. (2012) Innate immunity mediates follicular transport of particulate but not soluble protein antigen. J. Immunol. 188, 3724-3733. doi: 10.4049/jimmunol.1103312
Liu, X., Sun, Q., Wang, H., Zhang, L., Wang, J.Y. (2005) Microspheres of corn protein, zein, for an ivermectin drug delivery system. Biomaterials 26, 109-115. doi:10.1016/ j.biomaterials.2004.02.013
Lohcharoenkal, W., Wang, L., Chen, Y.C., Rojanasakul, Y. (2014) Protein nanoparticles as drug delivery carriers for cancer therapy. Biomed. Res. Int. 2014: 180549. doi: 10.1155/2014/180549
Saberianfar, R., and Menassa, R. (2017) Protein bodies: how the ER deals with high accumulation of recombinant proteins. Plant Biotechnol. J. 15, 671-673. doi: 10.1111/ pbi.12730
