Microarrays of natural and remodelled glycans
SUPERVISOR: IAIN WILSON
Background.
.Glycans are involved in a wide-range of cell-cell and host-parasite interactions; determining the natural interactions of glycans with receptors is a challenge as many important glycan ligands are only available in low quantities from natural sources. Also, current glycan arrays focus on mammalian glycans and not on those from non-mammalian systems of biotechnological or immunological relevance.
Recently, we have developed a new linker for non-reductive conjugation of glycans in order to facilitate their purification and immobilisation; these conjugates were printed and probed with lectins and antibodies (Jimenez-Castells et al., 2016). We have also used the AEAB label (i.e., 2-amino(N-aminoethyl)benzamide) to reductively conjugate N-glycans natural sources and probe these not only with lectins and sera of infected animals, but also with pentraxins (e.g., Hykollari et al., 2018).
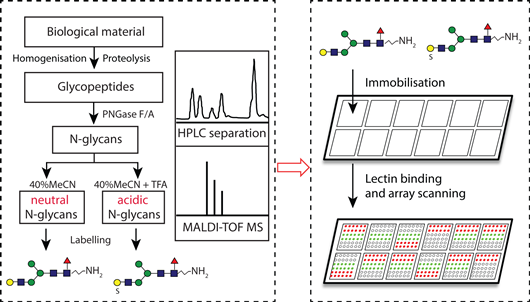
Aims and methods.
Primarily, we want to expand our range of glycans and glycan pools which we can print as well as test further antibodies and lectins with a focus on relevance to the human immune system. Mammalian N-/O-glycans can be released with either PNGase, hydrazine or household bleach and as necessary remodelled in vitro using glycosidases or glycosyltransferases to yield ‘unnatural’ fucosylated, GalNAc-modified or desialylated forms (cf. Iskratsch et al., 2009); non-mammalian N-glycans will be released with PNGase F or Ar or hydrazine (Yan et al, 2018), glycosaminoglycan-like chains by hydrazine (Vanbeselaere et al., 2018) and glycan moieties of non-mammalian glycolipids released by glycoceramidase or bleach; the bacterial polysaccharides isolated, where appropriate, after deacylation to release fatty acid moieties or after hydrofluoric acid to break phosphodiester bonds; the short oligosaccharides can be remodelled to generate NeuAc/NeuGc-modified or fucosylated forms (Jimenez-Castells et al., 2016).
These glycans will be derivatised either reductively or non-reductively with a fluorescent linker (such as AEAB), purified by HPLC and non-contact printed onto NHS-derivatised glass slides; where only small amounts of natural glycans are available, glycome pools rather than fractions will be employed. These can then be probed with (i) anti-carbohydrate antibodies, (ii) pentraxins (such as human serum amyloid P; see Hykollari et al, 2018), (iii) standard commercial lectins, (iv) fungal and human galectins and C-type lectins (such as CGL2, MBL, DC-SIGN available from either commercially or from collaborators; e.g., Butschi et al, 2010). The result is an unprecedented varied array of glycans to be probed with a variety of proteins and will expand our knowledge about which glycans are ligands for various antibodies and lectins, highlighting epitopes of immunological relevance or improving the basis for lectin-based screens in non-mammalian systems.
Butschi, A., Titz, A., Wälti, M.A., Olieric, V., Paschinger, K., Nöbauer, K., Guo, X., Seeberger, P.H., Wilson, I.B.H., Aebi, M., Hengartner, M.O., Künzler, M. (2010) Caenorhabditis elegans N-glycan core β-galactoside confers sensitivity towards nematotoxic fungal galectin CGL2. PLOS Pathogens 6, e1000717; doi: 10.1371/journal.ppat.1000717
Hykollari, A., Malzl, D., Eckmair, B., Vanbeselaere, J., Scheidl, P., Jin, C., Karlsson, N.G., Wilson, I.B.H., Paschinger, K. (2018) Isomeric Separation and Recognition of Anionic and Zwitterionic N-glycans from Royal Jelly Glycoproteins. Mol Cell Proteomics. 17, 2177-2196. doi: 10.1074/mcp.RA117.000462.
Iskratsch, T., Braun, A., Paschinger, K., Wilson, I.B.H. (2009) Specificity analysis of lectins and antibodies using remodelled glycoproteins. Anal. Biochem. 386, 133-4. doi: 10.1016/j.ab.2008.12.005
Jimenez-Castells, C., Stanton, R., Yan, S., Kosma, P., Wilson, I.B.H. (2016) Development of a multifunctional aminoxy-based fluorescent linker for glycan immobilization and analysis. Glycobiology 26, 1297-1307; doi: 10.1093/glycob/cww051
Vanbeselaere, J., Yan, S., Joachim, A., Paschinger, K., Wilson, I.B.H. (2018) The parasitic nematode Oesophagostomum dentatum synthesizes unusual glycosaminoglycan-like O-glycans. Glycobiology, in press. doi: 10.1093/glycob/cwy045
Yan, S., Vanbeselaere. J., Wöls, F., Jin, C., Blaukopf, M., Wilson, I.B.H., Paschinger, K. (2018). Core richness of N-glycans of Caenorhabditis elegans: a case study on chemical and enzymatic release. Anal Chem. 90, 928-935. doi: 10.1021/acs.analchem.7b03898
