Resolving dynamic protein conformations in multidomain enzymes with SAXS
SUPERVISORS: Roland LUDWIG and Rupert TSCHELIESSNIG
Background.
Small-angle X-ray scattering (SAXS) is a versatile method to obtain medium-resolution structures of proteins without the need for labelling or crystallisation. It also allows the determination of protein conformations in solution under different pH or solute conditions. In combination with molecular modelling techniques it can be used to assemble high-resolution structures of individual domains resolved by X-ray crystallography into a macromolecular complex or to determine the conformation of multidomain proteins [1, 2, 3]. Such multidomain proteins are commonly observed in the secretomes of biomass-degrading fungi, where multiple functionalities are required to bind to and process solid biopolymers.
Our enzyme of interest is cellobiose dehydrogenase (CDH). CDH features two mobile domains: (i) a carbohydrate binding module to bind onto cellulose microfibrils and (ii) a cytochrome domain to establish electron transfer with lytic polysaccharide monooxygenase (LPMO) for oxidative cellulose depolymerisation [4, 5, 6]. Small-angle scattering experiments have been performed to elucidate CDH’s conformation in solution already and proof the feasibility of the approach. The reported observations, however, are contradictory, thus no definite conclusions could be drawn so far [5, 7, 8, 9]. Studies on the most relevant conformation of CDH – when bound onto its substrate cellulose – have not even been started.
This project aims to: (i) elucidate the conformations of CDH in solution for different pH values and reduction and oxidation states of the cofactors, which are relevant for LPMO interaction studies, (ii) study the domain interaction of CDH and LPMO by a time-resolved technique, and (iii) determine the conformation of CDH bound onto cellulose. The project will be based on CDH from Crassicarpon hotsonii (PDB ID: 4QI6) and Neurospora crassa (PDB ID: 4QI7) from which the first crystal structures of full-length CDHs were obtained [5] and on N. crassa LPMO9C (PDB ID: 4D7U) [10]. 4QI6 represents CDH in its closed-state conformation, whereas 4QI7 was crystallized in the open-state. All SAXS experiments will be tested against open-state and closed-state conformers of CDH as definitive references, which will be generated by protein engineering and chemical cross-linking.
SAXS experiments to study the conformation of both enzymes (i) under different physiological conditions in solution, (ii) bound to cellulose, and (iii) their interaction will be performed at the Austrian SAXS beamline at the Synchrotron of Trieste. Cryo-SAXS will be implemented for time-resolved experiments of the cytochrome domain mobility by freeze-quenching the samples and then performing scattering experiments at the cryo-SAXS beamline at the Synchrotron of Trieste.
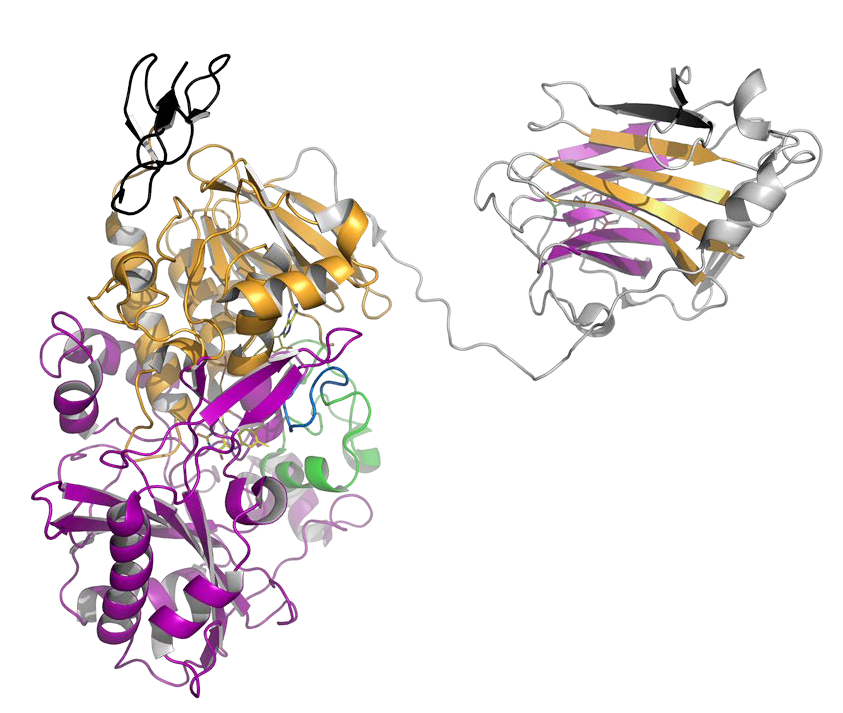
Cellobiose dehydrogenase is a carbohydrate oxidase with a flexible cytochrome domain. The mobility of this domain is important for its application in biosensors and biofuel cells. Fundamental factors affecting its mobility will be studied by using SAXS and cryoSAXS methods.
Aims and Methods.
In this project, the BioToP doctoral candidate will study the conformation of CDH in solution and bound to cellulose by SAXS and use the obtained experimental data for molecular models and simulations of the electron transfer between CDH and LPMO. The project aims elucidate the conformation and interaction of the involved domains. The obtained results are important to optimise biomass hydrolysis and biofuel production and to develop LPMO-based biosensors for analytical applications.
Two different CDHs from C. hotsonii (syn. Myriococcum thermophilum, ChCDHIIA) and N. crassa (NcCDHIIA) and an LPMO from N. crassa (NcLPMO9C) will be recombinantly produced and chromatographically purified for this project. Appropriate reference samples such as single CDH and LPMO domains and CDH fixed in the open-state (chimeric enzymes build from the domains of different CDHs) and in the closed-state (by chemical-cross linking of the cytochrome domain to the dehydrogenase domain) will also be produced according to existing protocols [11].
The PhD candidate will perform SAXS experiments in solution and determine the preferences of the closed- and open-state conformation of CDH in regard to pH, ion concentration and oxidised/reduced cofactors. SAXS experiments will be performed at the Austrian SAXS beamline at the synchrotron of Trieste in a flow-through quartz capillary, which is connected to a chromatographic system. This allows to precisely adjust ion concentration and pH. The domain conformation of CDH at the selected conditions and the impact of reduced or oxidised cofactors will be monitored by SAXS. The proposed flow/stopped-flow experimental setup is capable to initiate, support and monitor complex reaction kinetics and can be operated on-site.
The conformation of cellulose-bound enzymes will be studied with the same experimental setup, but using a cellulose-coated quartz capillary. Since the interaction and electron transfer between CDH and LPMO is way too fast for conventional SAXS, we will apply freeze-quenching together with cryo-SAXS (collaborator: OBINGER). Cryo-SAXS is a technique that is very well suited to determine highly radiation-sensitive proteins such as metalloenzymes [13]. We will analyse freeze-quenched samples prepared with the help of collaborators or, if possible, try to establish freeze-quenching on site to minimize the time and troubles of transporting frozen samples to the X-ray source.
The interaction of the domains based on the SAXS results will be studied at more detail by docking and molecular dynamics simulations (collaborator: OOSTENBRINK). Protein interactions and electron transfer between proteins will be measured in solution by fast kinetic methods (stopped-flow spectroscopy) and freeze-quenching (collaborator: OBINGER)
Collaborations within this thesis include OOSTENBRINK and OBINGER
Horejs, C., Pum, D., and Sleytr, U. B., Tscheliessnig, R. (2008) Structure prediction of an S-layer protein by the mean force method. J. Chem. Phys. 128:065106 doi:1.2826375
Grishkovskaya, I., Paumann-Page, M., Tscheliessnig, R., Stampler, J., Hofbauer,S., Soudi, M., Sevcnikar, B., Oostenbrink, C., Furtmüller, P.G., Djinović-Carugo K., Nauseef, W.M., Obinger, C. (2017) Structure of human promyeloperoxidase (proMPO) and the role of the propeptide in processing and maturation. J. Biol. Chem. 292:8244–8261. doi: 10.1074/jbc.M117.775031
Horejs, C., Gollner, H., Pum, P., Sleytr, U. B., Peterlik, H., Jungbauer, A., and Tscheliessnig, R. (2011) Atomistic structure of monomolecular surface layer self-assemblies: toward functionalized nanostructures. ACS Nano 5:2288–2297, doi: 10.1021/nn1035729
Harreither, W., Sygmund, C., Augustin, M., Narciso, M., Rabinovich, M.L., Haltrich, D., Ludwig, R. (2011) Catalytic properties and classification of cellobiose dehydrogenases from ascomycetes. Appl. Environ. Microbiol. 77: 1804–1815. doi: 10.1128/AEM.02052-10
Tan, T.-C., Kracher, D., Gandini, R., Sygmund, C., Kittl, R., Haltrich, D., Hallberg, B.M., Ludwig, R., Divne, C. (2015) Structural basis for cellobiose dehydrogenase action during oxidative cellulose degradation. Nat. Commun. 6:7542. doi: 10.1038/ncomms8542
Kracher, D., Scheiblbrandner, S., Felice, A.K.G., Breslmayr, E., Preims, M., Ludwicka, K., Haltrich, D., Eijsink, V.G.H., Ludwig, R. (2016) Extracellular electron transfer systems fuel cellulose oxidative degradation. Science, 352:1098–1101. doi: 10.1126/science.aaf3165
Lehner, D., Zipper, P., Henriksson, G., Pettersson, G. (1996) Small-angle X-ray scattering studies on cellobiose dehydrogenase from Phanerochaete chrysosporium. Biochim. Biophys. Acta, 1293:161–169. doi: 10.1016/0167-4838(95)00245-6
Bodenheimer, A.M., O'Dell, W.B., Stanley, C.B., Meilleur, F. (2017) Structural studies of Neurospora crassa LPMO9D and redox partner CDHIIA using neutron crystallography and small-angle scattering. Carbohydr. Res. 448:200–204. doi: 10.1016/j.carres.2017.03.001
Bodenheimer, A.M., O'Dell, W.B., Oliver, R.C., Qian, S., Stanley, C.B., Meilleur, F. (2018) Structural investigation of cellobiose dehydrogenase IIA: insights from small angle scattering into intra- and intermolecular electron transfer mechanisms. Biochim. Biophys. Acta, 1862:1031–1039. doi: 10.1016/j.bbagen.2018.01.016
Borisova, A.S., Isaksen, T., Dimarogona, M., Kognole, A.A., Mathiesen, G., Varnai, A., Rohr, A.K., Payne, C., Sorlie, M., Sandgren, M., Eijsink, V.G.H. (2015) Structural and functional characterization of a lytic polysaccharide monooxygenase with broad substrate specificity. J. Biol. Chem. 290:22955–22969. doi: 10.1074/jbc.M115.660183
Ma, S., Preims, M., Piumi, F., Kappel, L., Seiboth, B., Record, E., Kracher, D., Ludwig, R. (2017) Molecular and catalytic properties of fungal extracellular cellobiose dehydrogenase produced in prokaryotic and eukaryotic expression systems. Microbial Cell Factories, 16:37. doi: 10.1186/s12934-017-0653-5.
Silva, L.G., Plewka, J., Lichtenegger, H., Dias‐Cabral, A.C., Jungbauer, A., Tscheliessnig, R. (2018) The pearl necklace model in protein A chromatography: molecular mechanisms at the resin interface. Biotechnol. Bioeng. 116:76–86. doi: 10.1002/bit.26843
Meisburger, S. P., Warkentin, M., Chen, H., Hopkins, J. B., Gillilan, R. E., Pollack, L., and Thorne, R. E. B. (2013) Breaking the radiation damage limit with cryo-SAXS. Biophys. J. 104:227–236. doi: 10.1016/j.bpj.2012.11.3817
