Engineering of a bispecific mAb recognizing an optimized epitope of IgE as well as CD3 – a novel strategy for fighting allergic diseases
JOINT-SUPERVISORS: Florian RÜKER and Fritz ALTMANN
Background.
IgE is the antibody class that is responsible for all allergic diseases. It is one of the most attractive targets for therapeutic interventions [1]. The clinically validated monoclonal antibody (mAb) omalizumab (Xolair®) is the prototype anti IgE therapeutic. It is widely used to treat IgE-mediated conditions such as severe allergic asthma bronchiale or chronic urticaria [2]. Omalizumab inhibits binding of soluble IgE to its receptor (FcεRI) on mast cells and basophils.
As an alternative strategy to treat allergic symptoms, bispecific mAbs are expected to gain increased attention, since such molecules are already well established in clinical use for the treatment of cancer. Bispecific T-cell engagers (BiTEs®) recruit the patients’ cytotoxic T cells to targeted malignant cells. The prototypic example for this strategy is blinatumomab (trade name Blincyto), a bispecific mAb that recognizes CD19 on B cells and CD3 on T cells, thus directing the killing potential of cytotoxic T cells to certain malignancies of the B cell lineage such as leukemias [3]. A number of novel bispecific mAbs of the BiTE type are currently under development ([4] and www.clinicaltrials.gov).
In contrast to established treatment strategies for allergy that target the IgE molecule, it is the aim of the current project to target the cells that produce IgE, thus lowering its level in the patients’ serum.
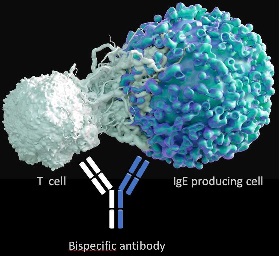 Initial attempts to eliminate IgE producing B cells have been described earlier using a chimeric T cell receptor approach [5] or vaccination approaches[6] or passive immunization strategies [7]. These yielded preliminary promising results, inspiring further research in the field. Kirak and Riethmüller [8] based their allergy-related research on the impressive clinical efficacy of blinatumomab and engineered a bispecific antibody molecule of the BiTE type that targets IgE on the one hand and CD3 on the other. Thus, this BiTE allowed the specific recognition of cells displaying IgE as a BCR, which include germinal center B cells and plasma cells, and in addition, IgE+ memory B cells. In the in vitro experiments described in their paper, the authors provide evidence for specific and efficient killing of cells that express IgE in the membrane bound form.
Initial attempts to eliminate IgE producing B cells have been described earlier using a chimeric T cell receptor approach [5] or vaccination approaches[6] or passive immunization strategies [7]. These yielded preliminary promising results, inspiring further research in the field. Kirak and Riethmüller [8] based their allergy-related research on the impressive clinical efficacy of blinatumomab and engineered a bispecific antibody molecule of the BiTE type that targets IgE on the one hand and CD3 on the other. Thus, this BiTE allowed the specific recognition of cells displaying IgE as a BCR, which include germinal center B cells and plasma cells, and in addition, IgE+ memory B cells. In the in vitro experiments described in their paper, the authors provide evidence for specific and efficient killing of cells that express IgE in the membrane bound form.
The mAb that was used by Kiran and Riethmüller is a fusion of an scFv derived from a murine anti-human IgE mAb and an scFv binding to CD3. Similar to Xolair®, the anti-IgE scFv recognizes an epitope on Cε3 and Cε4 domains of the IgE molecule that are described to be inaccessible on IgE when bound to its Fc receptors.
With the proposed project, we aim at the construction and characterization of alternative mAb constructs that are a “next generation” version to the one described above, and are improved in several critical aspects, making them better suited for possible therapeutic interventions. In particular, such a next generation mAb will have the following differentiating properties as compared to the one described earlier [9]:
A full size mAb with long in vivo half-life due to the presence of a binding site for FcRn
A mAb that is fully human in its sequence, thus reducing the risk of immunogenicity
A mAb that is optimized in its affinity towards IgE
Different mAbs with such properties, that recognize various epitopes located on IgE, thus allowing to select an epitope with superior efficacy as compared to the state of the art.
Research Questions:
Is there a way to construct a bispecific mAb directed against both, IgE and CD3, which is a human, or a humanized, or a chimeric mAb, has greatly reduced or zero risk of causing FceRI-crosslinking in treated patients, and has a long in vivo half-life?
- How to achieve heterodimerization of the heavy chains?
- How to achieve correct light chain pairing?
- Which particular clones of the two binding entities to choose?
- Is there a need for affinity maturation?
What are the biochemical, biophysical, immunological and biological characteristics of such a mAb?
- Sequence, structure and folding (CD spectroscopy, SEC, maybe even X-ray crystallography)
- Chain pairing and side products that may be formed due to chain mispairing (mass spectroscopy)
- Thermostability (DSC)
- Binding to the two antigens (ELISA, Biacore, Octet)
- Binding to FcRn
- Biological assays (specific killing of IgE displaying cells)
- Optimization of all these properties as needed
- Would such an mAb be a potential therapeutic for treating severe forms of allergy?
- Can we gain access to a suitable animal model?
Aims and expected outcomes.
To construct bispecific, heterodimeric, full-size mAbs that recognize different epitopes on IgE and that recognize CD3 on T cells and can activate these cells.
To select a mAb that recognizes an epitope on IgE that is positively differentiating as compared to the mAbs described in the literature.
To characterise such a mAb in molecular details, including its biochemical, biophysical, immunological and biological properties
To gain enhanced knowledge on the general design principles and how they influence the characteristics and the expressability of the mAb
To develop such a mAb to a point where a pharmaceutical company could be interested in further developing it to IND status and onwards to a Phase I clinical trial.
Project plan & methods.
A number of anti-IgE mAbs will be the starting point of the project. These test mAbs will be expressed as chimeric or humanized molecules, engineered into a bispecific format together with the humanized VH and VL domains of OKT3 and human constant regions for the light and the heavy chains.
Several ways of achieving heterodimerization of heavy chains have been described in the literature and have been reviewed recently [10], [11]. Based on experience in the lab with such formats the best suited one will be chosen as the basis for the various mAb constructs.
In order to achieve correct light chain pairing, two engineering strategies developed in our group will be tested and the better suited one will be chosen for the final mAb construct.
All mAb constructs will be expressed transiently in either HEK293 or ExpiCHO cells in order to produce sufficient material for characterization.
ELISA, Biacore and Octet will be used as methods for determining the binding of the bispecific mAb candidates to both, IgE and CD3. In addition, pH sensitive binding to FcRn will be assessed in order to make sure that the final construct has a long in vivo half-life in patients. In case it is deemed necessary, an affinity maturation of the IgE binding mAb will be performed.
Mass spectroscopy will be critically important for the characterization of the expressed mAb molecules, in order to asses and optimize:
- the correct chain pairing for heterodimerizing heavy chains
- the assembly of the heavy chains with their corresponding light chains
- bottlenecks in the biosynthesis of four polypeptide components
Testing the stability of the generated candidate mAbs by DSC will allow to guide our selection of heterodimerization and light chain pairing strategy.
Finally, a cell killing assay similar to the one described in [8] will be performed in order to assess the cytotoxic activity mediated by the bispecific mAb constructs. For this purpose, a Burkitt-Lymphoma- derived Ramos reporter cell line will be used.
Collaborations and time plan:
Construction and expression of the candidate mAbs will be performed in the group of RÜKER/WOZNIAK-KNOPP, and characterization will be done there and in collaboration with the Core Facility for Biomolecular and Cellular analysis (Irene SCHAFFNER). The group of Fritz ALTMANN will be responsible for mass spec analysis of the mAb candidates and to provide constant feedback in order to support their optimization.
It is envisaged that after the 1st year, the first set of constructs is characterized to a level where a second and optimized generation can be designed, which should then be accomplished after a further 6 months. At this point in time, biological assays will be set up and applied for the biological characterization of the constructs. In parallel, further optimization of the mAbs can be undertaken, e.g. to affinity mature the IgE binding antibody.
A stay abroad of 6 months is anticipated, possible labs for this stay will be identified in due course of the project.
[1] S. Eyerich, M. Metz, A. Bossios, and K. Eyerich, “New biological treatments for asthma and skin allergies,” Allergy, 2019.
[2] C. Incorvaia, M. Mauro, E. Makri, G. Leo, and E. Ridolo, “Two decades with omalizumab: What we still have to learn,” Biologics: Targets and Therapy. 2018.
[3] E. Curran and W. Stock, “Taking a ‘bite out of all’: Blinatumomab approval for MRD-positive ALL,” Blood, 2019.
[4] A. Krishnamurthy and A. Jimeno, “Bispecific antibodies for cancer therapy: A review,” Pharmacology and Therapeutics. 2018.
[5] J. Lustgarten and Z. Eshhar, “Specific elimination of IgE production using T cell lines expressing chimeric T cell receptor genes,” Eur. J. Immunol., 1995.
[6] S. Haba and A. Nisonoff, “Effects of syngeneic anti-IgE antibodies on the development of IgE memory and on the secondary IgE response,” J. Immunol., 1994.
[7] S. Feichtner et al., “Targeting the Extracellular Membrane-Proximal Domain of Membrane-Bound IgE by Passive Immunization Blocks IgE Synthesis In Vivo,” J. Immunol., 2008.
[8] O. Kirak and G. Riethmüller, “A novel, nonanaphylactogenic, bispecific IgE-CD3 antibody eliminates IgE+ B cells,” J. Allergy Clin. Immunol., 2015.
[9] S. Krah, H. Kolmar, S. Becker, and S. Zielonka, “Engineering IgG-Like Bispecific Antibodies—An Overview,” Antibodies, 2018.
[10] S. Dietrich et al., “Constant domain-exchanged Fab enables specific light chain pairing in heterodimeric bispecific SEED-antibodies,” Biochim. Biophys. Acta - Proteins Proteomics, 2019.
[11] M. Bönisch et al., “Novel CH1:CL interfaces that enhance correct light chain pairing in heterodimeric bispecific antibodies,” in Protein Engineering, Design and Selection, 2017.
