Design and engineering of switchable cellobiose dehydrogenase for implantable blood glucose biosensors
SUPERVISOR: Roland LUDWIG
Rationale.
For decades, one of the major visions in the field of biomedical diagnostics has been to develop long-term stable, implantable bioelectronics. Most notably, implantable blood glucose biosensors offer tremendous benefits to further improve monitoring and self-management of Diabetes patients (Figure 1). However, long-term operational stability of biosensors is mostly hampered by a lack of suitable biorecognition elements with high selectivity for glucose and long-term operational stability.
In my PhD project, I aim to develop next-generation glucose-sensing enzymes with improved properties to use as biorecognition elements for long-term stable implantable blood glucose biosensors. For this purpose, I will use a combination of rational design and directed evolution to design and engineer next-generation cellobiose dehydrogenase (CDH) with high glucose turnover, low disaccharide and oxygen cross-reactivity, and—most notably—switchable activity to significantly improve the long-term stability of the enzyme.
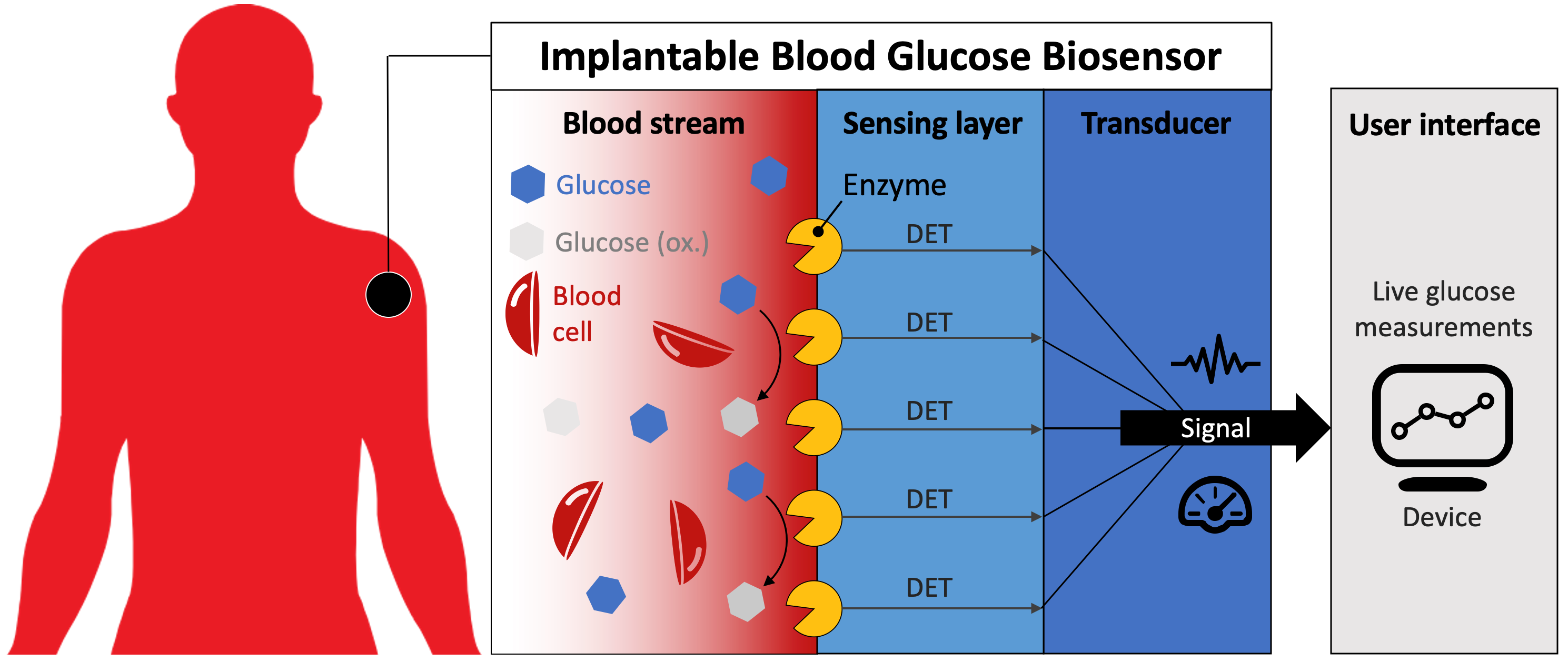
Figure 1 | Schematic of implantable blood glucose biosensor. DET: direct electron transfer.
Background.
The future of blood glucose monitoring—What are the future visions in biomedical diagnostics? How can we provide tools which control the glucose level of diabetic people at high quality, continuously, pain-free and over a long term to allow them to live a life similar to that of non-diabetic people? Long-term stable, implantable blood glucose biosensor devices provide a solution to these problems since they offer quick, easy, and reliable in vivo blood glucose measurements. However, such devices are still not yet commercially available for diabetic patients. For decades, the major challenge in developing such innovative devices has been the limited long-term operational stability of the biorecognition elements used. Hence, next-generation enzymes with long-term stability, high sensitivity for glucose, and low cross-reactivity to other carbohydrates are desperately needed.
Cellobiose dehydrogenase as a suitable biorecognition element—Cellobiose dehydrogenase (CDH) is a fungal flavocytochrome with a unique bipartite protein structure consisting of a catalytic active dehydrogenase (DH) domain connected via a flexible linker to a mobile cytochrome C (CYT) domain (Figure 2). This special dual structure enables CDH to catalyse the oxidation of glucose via its DH domain, and subsequently perform a direct electron transfer (DET) step using the mobile CYT domain. In this way, electrons are efficiently directed towards a terminal electron acceptor, without the need of an intermediary electron mediator. The capacity of CDH to perform DET renders it a promising candidate for incorporation as a biorecognition element (BRE) into implantable blood glucose biosensors.
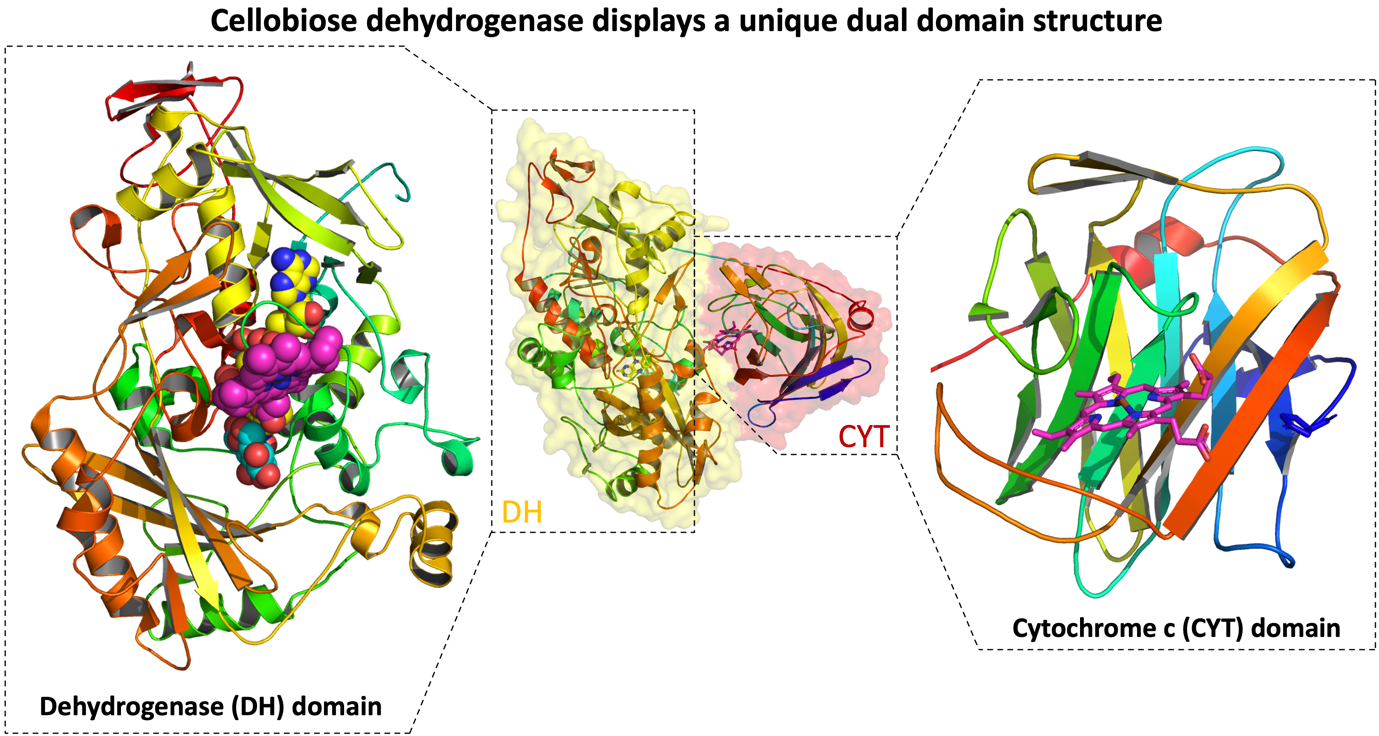
Figure 2 | Crystal structure of cellobiose dehydrogenase from Myriococcum thermophilum (PDB: 4QI6) displays the unique dual domain structure of CDH in the closed conformation. The domain interface of the DH (left) and CYT domain (right) is shown.
Objectives.
In my PhD project, I will develop next-generation cellobiose dehydrogenase (CDH) with improved properties to use as biorecognition elements for implantable blood glucose biosensors. There are, in particular, three highly desired properties:
- Improved glucose turnover rates to further enhance the signal intensity for precise and accurate detection of blood glucose levels
- Decreased cross-reactivity towards disaccharides to increase signal-to-noise ratios and overall sensitivity of the biosensor
- Suppressed oxygen reactivity to expand the long-term operational stability of the enzyme, even in highly enriched oxygen environments such as human blood
- Switchable activity for remote control and improved long-term stability of the implantable biosensor device (Figure 3)
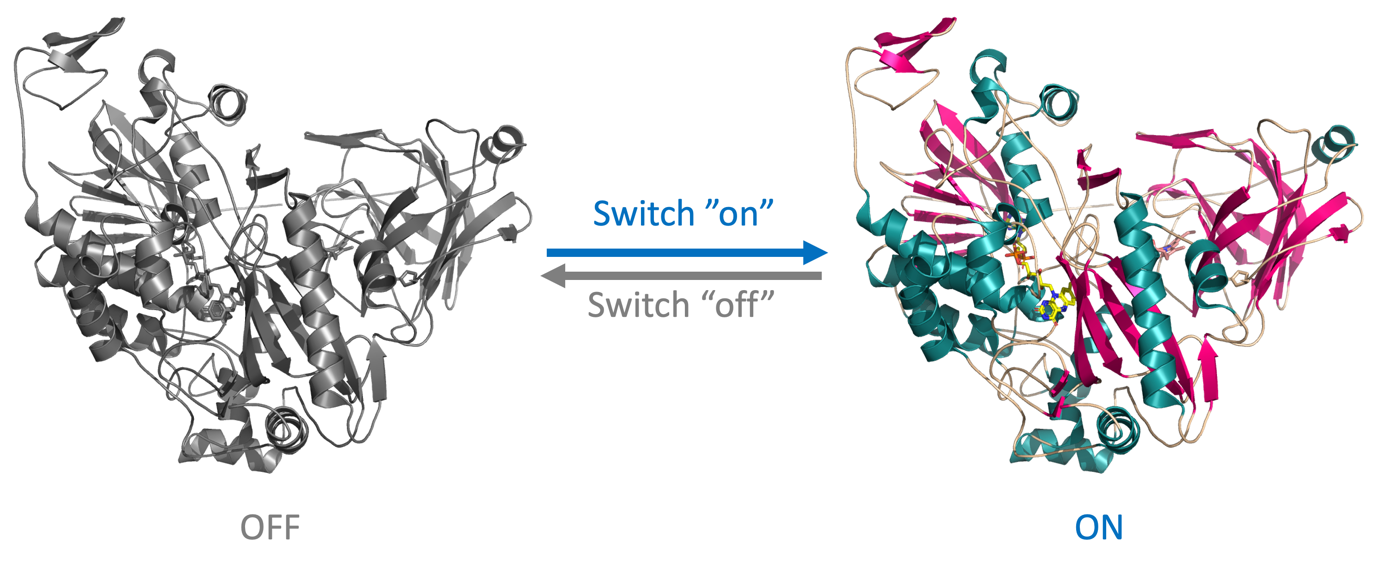
Figure 3 | Schematics of switchable CDH.
Methods.
To achieve this ambitious goal, I will use three consecutive strategies:
- First, I will investigate various new CDH genes from different fungal sources to identify hitherto unexplored variants with promising properties
- Secondly, I will develop next-generation variants with favorable kinetic constants for glucose and strict, oxygen defying properties by targeting key structural hotspots via a combination of rational design and directed evolution
- Thirdly, I will engineer CDH into a switchable enzyme with a controllable catalytic activity that can be tuned by external stimuli like ionic charges or the applied potential.
- Lastly, I will combine all of the described features into one next-generation CDH that can be subsequently used as a biorecognition element on implantable blood glucose biosensors.
Collaborations within this thesis include OOSTENBRINK and TSCHELIEßNIG
REFERENCES:
Bollella, P. and Gorton, L. (2018) ‘Enzyme based amperometric biosensors’, Current Opinion in Electrochemistry. Elsevier B.V., 10, pp. 157–173. doi: 10.1016/j.coelec.2018.06.003.
Bollella, P., Gorton, L. and Antiochia, R. (2018) ‘Direct electron transfer of dehydrogenases for development of 3rd generation biosensors and enzymatic fuel cells’, Sensors (Switzerland), 18(5). doi: 10.3390/s18051319.
Harding, J. L. et al. (2019) ‘Global trends in diabetes complications: a review of current evidence’, Diabetologia. Diabetologia, 62(1), pp. 3–16. doi: 10.1007/s00125-018-4711-2.
Kracher, D. et al. (2016) ‘Extracellular electron transfer systems fuel cellulose oxidative degradation’, Science, 352(6289), pp. 1098–1101. doi: 10.1126/science.aaf3165.
Ortiz, R. et al. (2017) ‘Engineering of Class II Cellobiose Dehydrogenases for Improved Glucose Sensitivity and Reduced Maltose Affinity’, ChemElectroChem, 4(4), p. 750. doi: 10.1002/celc.201700226.
Pérez, J. A. C. et al. (2019) ‘Bioinspired biomaterials and enzyme-based biosensors for point-of-care applications with reference to cancer and bio-imaging’, Biocatalysis and Agricultural Biotechnology. Elsevier Ltd, 17(October 2018), pp. 168–176. doi: 10.1016/j.bcab.2018.11.015.
Scheiblbrandner, S. and Ludwig, R. (2020) ‘Cellobiose dehydrogenase: Bioelectrochemical insights and applications’, Bioelectrochemistry. The Authors, 131, p. 107345. doi: 10.1016/j.bioelechem.2019.107345.
Tan, T. C. et al. (2015) ‘Structural basis for cellobiose dehydrogenase action during oxidative cellulose degradation’, Nature Communications, 6(May). doi: 10.1038/ncomms8542.
Zafar, M. N. et al. (2012) ‘Characteristics of third-generation glucose biosensors based on Corynascus thermophilus cellobiose dehydrogenase immobilized on commercially available screen-printed electrodes working under physiological conditions’, Analytical Biochemistry. Elsevier Inc., 425(1), pp. 36–42. doi: 10.1016/j.ab.2012.02.026.
