Development of a novel binder scaffold
SUPERVISOR: Christian OBINGER
Background.
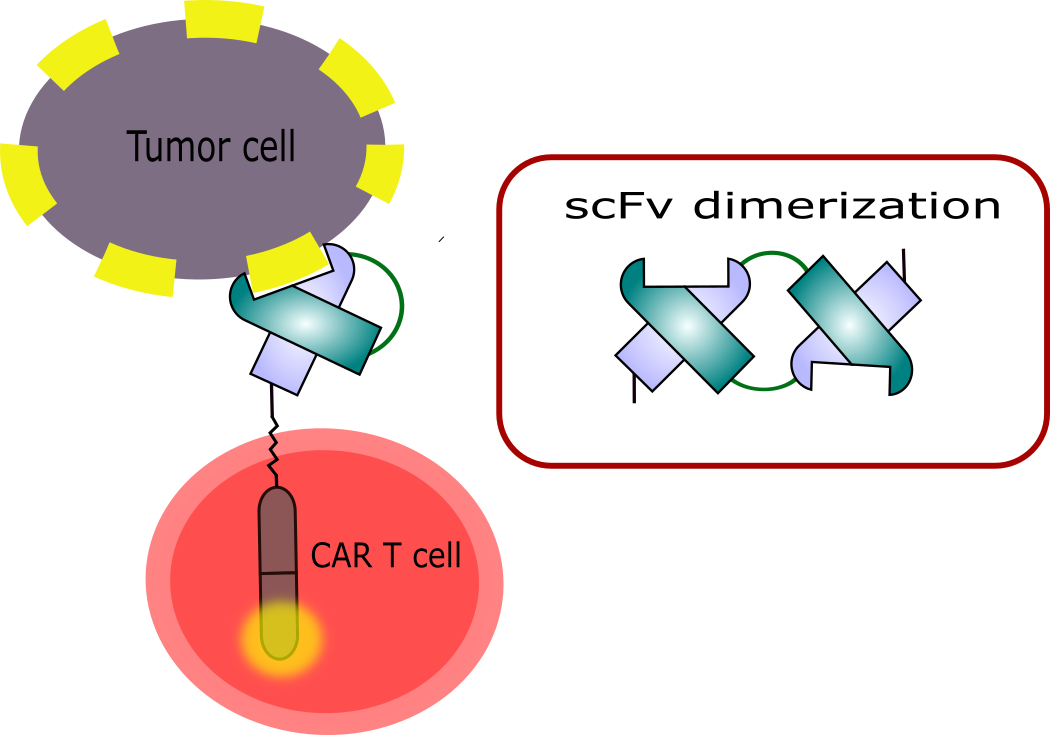
Adoptive immunotherapy with T cells expressing chimeric antigen receptors (CARs) has shown great success in the clinics (Batlevi, Matsuki et al.). As a consequence, two products - Yescarta® and Kymriah® - were authorized by the EMA in 2018. CARs are chimeric proteins carrying an antigen-binding domain (usually a fragment derived from an antibody), which is connected to an intracellular signaling domain, which will activate the T cell after antigen recognition and further trigger cytotoxic activity. In order to specifically target cancer cells, CAR T cells can be engineered to target a tumor associated antigen (TAA) expressed on the cell surface.
Several limitations are associated with the use of CAR T cells in the clinics including a condition known as cytokine release syndrome (CRS). Furthermore, the use of single chain variable fragments (scFvs) as antigen-binding unit bears the risk of receptor cross-pairing, which will provoke latent activation and tonic signaling of the genetically manipulated T cells.
In this project the risk of cross-pairing with other CAR molecules shall be addressed by replacing the commonly used scFv with an alternative binder scaffold. In the past, a charge-reduced version of the Sso7d protein derived from the hyperthermophilic organism Sulfolobus solfataricus (Traxlmayr, Kiefer et al. 2016)has been tested for this purpose. Still, in order to enable the application of CAR T cells in the clinic, a novel, human binder scaffold shall be developed within this project. Protein stability has previously been shown to be a crucial prerequisite for successful protein engineering and affinity maturation (Bloom, Labthavikul et al. 2006). Therefore, a thermostable structure will be chosen as a scaffold for molecular antigen recognition.
Aims and methods.
The aim of this project is the development and affinity maturation of a novel binder scaffold.
Yeast surface display will be used to identify binders against several tumor associated antigens (TAA). Next, the selected binders will be solubly expressed and characterized with regard to their thermostability using differential scanning calorimetry (DSC). Further, the selected binders shall be assessed regarding the formation of aggregates by size-exclusion chromatography (SEC).
The most promising binder scaffold shall further be assessed with regard to its binding affinity to the respective TAA by using surface plasmon resonance (SPR). In order to investigate whether the use of alternative scaffolds provides a valuable alternative to scFvs the most interesting construct will be cloned into a CAR backbone and tested for its ability to activate T cells in response to TAA-positive target cells. T cell activation will be characterized in vitro by measuring degranulation based on FACS analysis of CD107a surface expression, release of interferon-gamma and cytolysis of target cells using a luciferase-assay (LEHNER).
REFERENCES:
Batlevi, C. L., et al. (2016). "Novel immunotherapies in lymphoid malignancies." Nature Reviews Clinical Oncology 13(1): 25-40.
Bloom, J. D., et al. (2006). "Protein stability promotes evolvability." Proceedings of the National Academy of Sciences 103(15): 5869-5874.
Traxlmayr, M. W., et al. (2016). "Strong Enrichment of Aromatic Residues in Binding Sites from a Charge-neutralized Hyperthermostable Sso7d Scaffold Library." Journal of Biological Chemistry 291(43): 22496-22508.
