Fundamentals for ultrafast virus analytics
PRINCIPAL INVESTIGATOR: Alois JungbauerBackground.
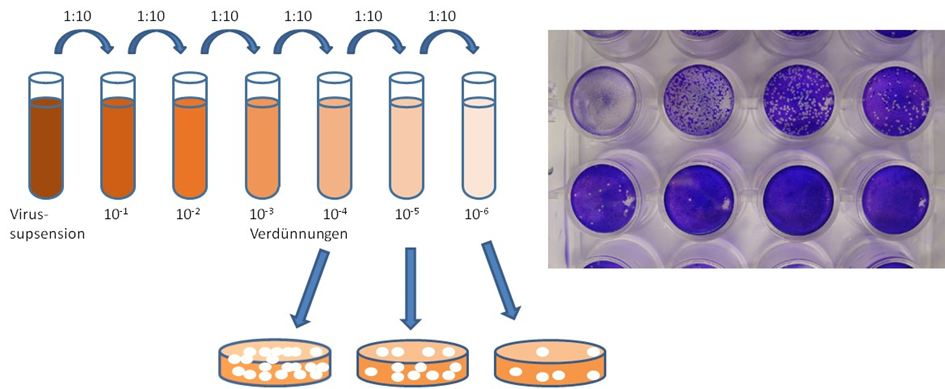
The determination of infectivity in cell culture tests usually takes five days to a week. The tests are error-prone and the result is often operator-dependent, i.e. different depending on the person performing them. These tests complicate any process development, release or general research activities in the context, e.g. development of vaccines or gene therapy vectors based on viruses. The fundamentals for a novel ultrafast method for determination of infectivity of virus will be generated. As model virus Baculovirus and Measles virus will be used. Measles are a general vector for vaccination. Baculovirus is a vector for production of proteins in insect cells.
Aims and methods.
Three different analytical methods are being developed in parallel: (1) measurement of the Cy-toslic Ca ++ shift after infection of indicator cell lines with our model viruses (2) triggering of an "early interferon / immune response" after infection of indicator cell cells with the model viruses and (3) measurement of certain ones mRNA sequences after the model viruses have propagated in the indicator cell lines. In addition to the infectivity, the particle content must also be measured, as a bioreactor harvest contains a number of particles that are very similar to the viruses, but cannot really be separated in the following cleaning processes (the health authorities not only require the information about new vaccines Infectivity but also via the particle size distribution and the content of VLPs). With nanoparticle tracking analysis, field flow fractionation, preparative size exclusion chromatography and flow cytometry with specific antibodies against surface proteins, we will therefore conduct research on particle content measurement.
Barczak, Wojciech et al. “Universal Real-Time PCR-Based Assay for Lentiviral Titration.” Molecular Biotechnology 57 (2015): 195–200.
Wang et al. “A reliable and feasible qPCR strategy for titrating AAV vectors” Med Sci Monit Basic Res (2013); 19:187-193
Zang Y, et al. „Development of one-step real-time PCR assay for titrating trivalent live attenuated influenza vaccines”. Human Vaccines & Immunotherapeutics. (2014)10(12):3642-3648.
Ammour et al “Quantification of measles, mumps and rubella viruses using real-time quantitative TaqMan-based RT-PCR assay”(2013) Journal of Virological Methods, Volume 187, 1,
Schalk, et al. “Estimation of the number of infectious measles viruses in live virus vaccines using quantitative real-time PCR (2004) Journal of Virological Methods, Volume 117, Issue 2, ,
Steppert, P., et al . Quantification and characterization of virus-like particles by size-exclusion chromatography and nanoparticle tracking analysis (2017) Journal of Chromatography A, 1487, pp. 89-99.
Steppert, P.,et al. Separation of HIV-1 gag virus-like particles from vesicular particles impurities by hydroxyl-functionalized monoliths (2017) Journal of Separation Science, 40 (4), pp. 979-990.
Steppert, et al. Purification of HIV-1 gag virus-like particles and separation of other extracellular particles (2016) Jour-nal of Chromatography A, 1455, pp. 93-101.
Pereira Aguilar, et al. At-line multi-angle light scattering detector for faster process development in enveloped virus-like particle purification (2019) Journal of Separation Science 42(16), pp. 2640-2649
