Glycobiology aspects of Tannerella serpentiformis
SUPERVISOR: Christina SCHÄFFER
Background.
Periodontitis is a global, multifactorial inflammatory biofilm disease for which no targeted treatment is currently available; it crucially involves Tannerella forsythia (Tf). Protein O-glycosylation and N-acetylmuramic acid auxotrophy are key aspects in the bacterium’s pathogenicity and multispecies biofilm lifestyle. Tf employs a unique general protein glyco-sylation system targeting the abundant S-layer [1] and other virulence proteins; a nonulosonic acid is crucial for the interaction with the host and the bacterium’s establishment in the biofilm community [2]. The genome sequences of periodontitis-associated Tf strains reveal the presence of a distinct O-glycosylation gene cluster [3], while the genome of the novel, non-pathogenic bacterium Tannerella serpentiformis (Tser) [4,5], which is the closest cultivated phylogenetic relative to Tf, has a different glycosylation gene cluster inserted between the conserved genes for a Wzx flippase and a GtfE fucosyltransferase, but lacks nonulosonic acid biosynthesis genes. This FucT is conserved in the Bacteroidetes phylum and might be pivotal glycan biosynthesis. Tser has the genetic make-up for synthesizing the essential peptide-glycan component N-acetylmuramic acid, which might confer a colonization advantage to Tser.
We hypothesize that an altered glycome and peptidoglycan sugar metabolism contribute to the differ-ence of periodontal pathogenesis by Tser and provide a new foundation to further understand the genome evolution and mechanisms of bacte-ria-host interaction in closely related oral microbes with different pathogenicity potential.
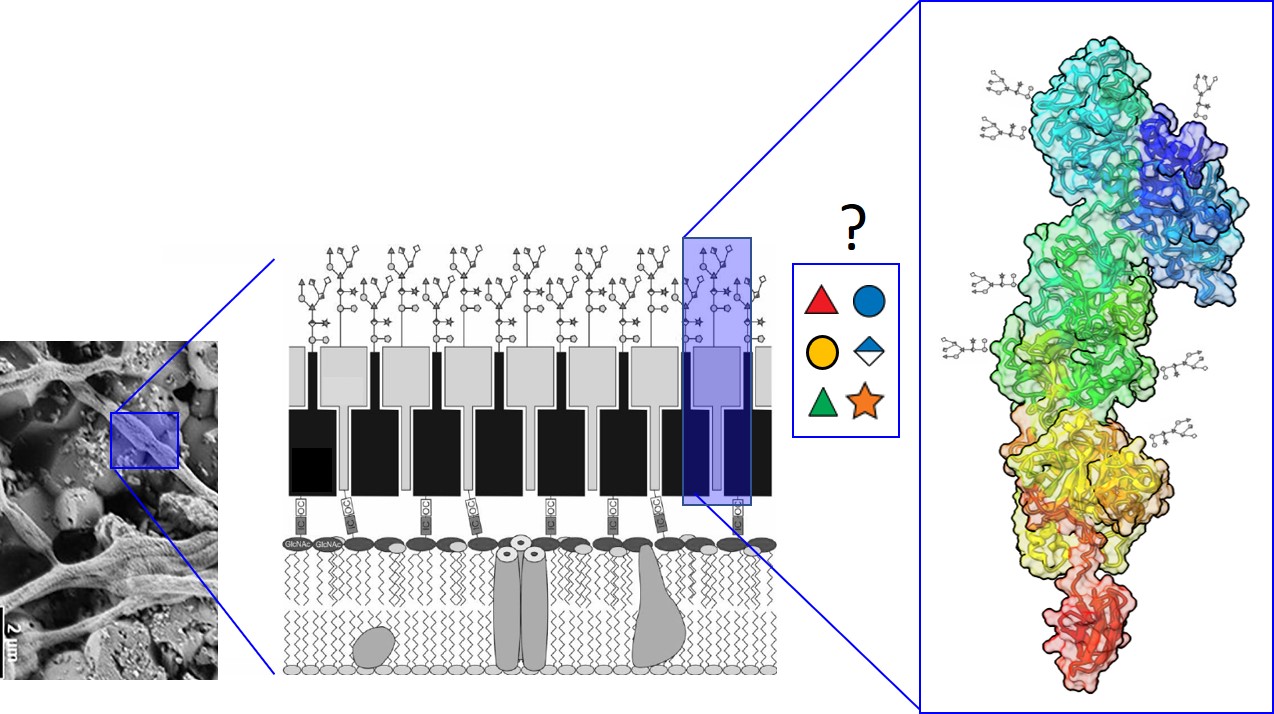
Aims and methods.
We will investigate protein O-glycosylation in Tser exemplified by the bacterium’s abundant S-layer proteins and delineate possible implications for the bacterium's lifestyle and host interaction. Methods include anaerobic bacterial cultivation, MS and NMR for glycan structure elucidation, glycoproteomics, MD modelling and simulation of S-layer glyco-proteins, biofilm techniques, imaging, genetic manipulation, and immune response profiling.
Experimental setup1. S-layer glycopeptides will be prepared and O-glycans will be rleased by reductive β-elimination [1]. Once the structure of the S-layer protein O-glycan has been elucidated by NMR spectroscopy (cooperation H. KÄHLIG) it will be sought to determine a relationship between the structure and the glycosylation gene cluster. Specifically, the role of the conserved FucT will be assessed by gene knock-out and in cross-complementation studies involving Tf and P. gingivalis. The enzyme will be characterized in vitro with an established assay and subjected, together with its available orthologous to X-ray crystallography (cooperation: S. EVANS).
2. The glycosylation potential of Tser will be assessed by glycoproteomics (cooperation: J. STADLMANN).
3. MD modelling and simulation of the two S-layer glycoproteins TssA and TssB, which build up a 2D-crsytalline monolayer on the bacterial cell surface will be performed (cooperation: C. OOSTENBRINK, A. BONVIN); the S-layer will be imaged on intact cells using freeze-etching (cooperation: T. HEUSER).
4. Biofilm growth of Tser will be assessed in mono-and multispecies settings with and without provision of N-acetylmuramic acid, including analysis of biofilm structure and outcompeting of pathogenic Tf and P. gingivalis (qPCR, FISH) [6].
5. Time permitting, to determine if and to which extent protein glycosylation as a single factor can affect the pathogenicity potential of different Tannerella species, glycosylation information between Tser and Tf shall be exchanged and the immune response of select host cells important to periodontitis analyzed (cooperation: O. ANDUKHOV).
Collaborations.
Collaborations within this thesis will include: STADLMANN (MS, glycoproteomics, BOKU); KÄHLIG (NMR; NMR Center, Univ. Vienna), OOSTENBRINK (MD simulation and modelling, BOKU) HEUSER (EM facility, Vienna Biocenter), EVANS (X-ray crystallography, Uni. Victoria, Canada), ANDRUKHOV (immunology; Med. Uni. Wien);
1. Posch G. et al. (2011) Characterization and scope of S-layer protein O-glycosylation in Tannerella forsythia. J Biol Chem 286, 38714-38724. doi:10.1074/jbc.M111.284893
2. Bloch S et al. (2019) Nonulosonic acids contribute to the pathogenicity of the oral bacterium Tannerella forsythia. Interface Focus 9, 20180064. doi:10.1098/rsfs.2018.0064
3. Tomek M.B. et al. (2018) A general protein O-glycosylation gene cluster encodes the species-specific glycan of the oral pathogen Tannerella forsythia: O-glycan biosynthesis and immunological implications. Front Micirobiol 9, 2008. doi:10.3389/fmicb.2018.02008
4. Ansbro K. et al. (2020) Tannerella serpentiformis sp. nov., isolated from the human mouth. Int J Syst Evol Microbiol 70, 3749-3754. doi:10.1099/ijsem.0.004229
5. Frey A.M. et al. (2018) Characterisation and pure culture of putative health-associated oral bacterium BU063 (Tannerella sp. HOT-286) reveals presence of a potentially novel glycosylated S-layer. FEMS Microbiol Lett 365, fny180. doi:10.1093/femsle/fny180
6. Bloch S. et al. (2017) Behavior of two Tannerella forsythia strains and their cell surface mutants in multispecies oral biofilms. Mol Oral Microbiol 32, 404-418. doi:10.1111/omi.12182
