Elucidating the molecular mechanism of engineered binders targeting the activated conformation of EGFR
PRINCIPAL INVESTIGATOR: Michael TRAXLMAYR
Background.
The epidermal growth factor receptor (EGFR) is member of the ErbB family of receptor tyrosine kinases. In its inactive state, EGFR is predominantly present as a monomer with a tethered extracellular domain. Upon activation by a ligand, its extracellular domain switches to an open, extended conformation, which triggers dimerization and activation of the intracellular signaling domains (Kovacs et al., 2015). As a consequence, Tyr residues on the C-terminal tail of EGFR are phosphorylated, which triggers intracellular signaling pathways such as the MAPK- and the PI3K-pathway. Given its central role in transmitting proliferative and survival signals across the plasma membrane, it is not surprising that EGFR signaling is frequently upregulated in human cancers. One frequently observed mechanism of constitutive EGFR activation in human tumors is amplification of the EGFR gene and subsequent EGFR overexpression. We and others have observed that EGFR overexpression leads to constitutive, ligand-independent EGFR signaling (Endres et al., 2013; Wagner et al., 2022).
To specifically target cancer-associated EGFR molecules, we have engineered artificial binding domains that specifically recognize the activated conformation of EGFR (Dobersberger et al., paper in preparation). These binding domains show strongly increased affinities towards EGFR in the presence of any of its ligands (e.g. EGF or TGF-α), demonstrating that they recognize the extended, activated conformation of EGFR irrespectively of the type of bound ligand (EGF or TGF-α).
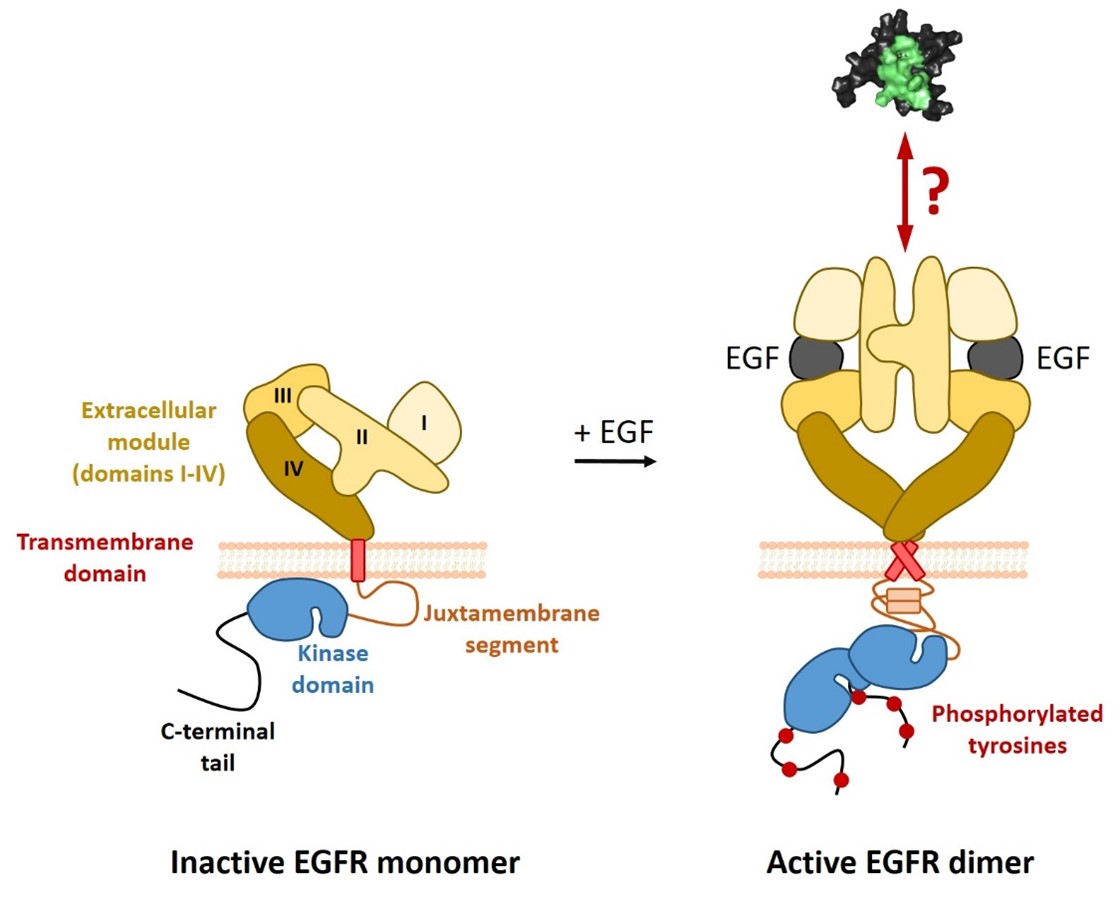
Figure adapted from Wagner et al. (Wagner et al., 2021).
Aims and methods.
It is the goal of this project to elucidate the molecular mechanism of these engineered binding domains, i.e. how they specifically recognize the activated conformation of EGFR. This will yield valuable information for engineering future generations of conformation-specific binders for more specific targeting of human tumors.
First, we will aim to solve the crystal structure of the engineered binders in complex with the activated extracellular domain of EGFR. As a complementary approach, we will also create a library of EGFR variants using error prone PCR and express them on the surface of yeast using protocols that are well-established in our laboratory (Traxlmayr et al., 2012; Zajc et al., 2020; Zajc et al., 2022). This yeast-displayed EGFR library will subsequently be selected for maintained folding (using conformationally specific antibodies) and for loss-of-binding to the engineered binding domains. Thus, we will select for EGFR variants that possess a native conformation, but which lost their ability to bind to the engineered binders, thus yielding mutations in the binding epitope. Together, crystallography and yeast display-guided epitope mapping will yield valuable information on the molecular recognition of activated EGFR.
Furthermore, the engineered binding domains will be analyzed with respect to their tumor specificity and affinity by performing titration experiments on a comprehensive panel of EGFR-positive or –negative human cell lines in the presence or absence of a range of EGFR ligands. Finally, based on their biochemical characteristics, the most promising binders will be chosen for CAR T cell experiments, where they will serve as CAR antigen binding domains.
Collaborations within this thesis will include Manfred LEHNER, CCRI, Vienna (CAR T cells).
Endres, N.F., Das, R., Smith, A.W., Arkhipov, A., Kovacs, E., Huang, Y., Pelton, J.G., Shan, Y., Shaw, D.E., Wemmer, D.E., et al. (2013). Conformational coupling across the plasma membrane in activation of the EGF receptor. Cell 152, 543-556.
Kovacs, E., Zorn, J.A., Huang, Y., Barros, T., and Kuriyan, J. (2015). A structural perspective on the regulation of the epidermal growth factor receptor. Annu Rev Biochem 84, 739-764.
Traxlmayr, M.W., Hasenhindl, C., Hackl, M., Stadlmayr, G., Rybka, J.D., Borth, N., Grillari, J., Ruker, F., and Obinger, C. (2012). Construction of a stability landscape of the CH3 domain of human IgG1 by combining directed evolution with high throughput sequencing. J Mol Biol 423, 397-412.
Wagner, A., Galicia-Andres, E., Teufl, M., Gold, L., Obinger, C., Sykacek, P., Oostenbrink, C., and Traxlmayr, M.W. (2022).
Identification of Activating Mutations in the Transmembrane and Extracellular Domains of EGFR. Biochemistry 61, 2049-2062.
Wagner, A., Teufl, M., Gold, L., Lehner, M., Obinger, C., Sykacek, P., and Traxlmayr, M.W. (2021). PhosphoFlowSeq - A High-throughput Kinase Activity Assay for Screening Drug Resistance Mutations in EGFR. J Mol Biol 433, 167210.
Zajc, C.U., Dobersberger, M., Schaffner, I., Mlynek, G., Puhringer, D., Salzer, B., Djinovic-Carugo, K., Steinberger, P., De Sousa Linhares, A., Yang, N.J., et al. (2020). A conformation-specific ON-switch for controlling CAR T cells with an orally available drug. Proc Natl Acad Sci U S A 117, 14926-14935.
Zajc, C.U., Teufl, M., and Traxlmayr, M.W. (2022). Affinity and Stability Analysis of Yeast Displayed Proteins. Methods Mol Biol 2491, 155-173.
