Fast and reproducible generation of stable mammalian cells by baculovirus mediated genome integration
SUPERVISOR: Reingard GRABHERR
Background.
Efficient recombinant protein production requires stable cell lines or often relies on inefficient transfection processes. Baculoviral transduction of mammalian cells (BacMam) offers cost-effective and robust gene transfer and straightforward scalability. The advantages over conventional approaches are, no need of high biosafety level laboratories, efficient transduction of various cell types and transfer of large transgenes into host cells. In our study, we aim to develop a high expression cassette to increase yields of baculoviral transduction. The establishment follows a sequential approach by first identifying the strongest promoter, followed by intron and WPRE sequences as enhancer elements for transcription and translation. The resulting REMBAC-cassette was compared to conventional transfection in suspension and adherent cells. Irrespective of the cell line, transduction reached nearly 100% efficiency and led to almost 10-fold increases of gene expression levels. We confirmed these results in larger scale with batch and fed-batch cultivations. Finally, expression of different soluble proteins with high degrees of complexity and confirmed the versatility of our established cassette. Overall, the REMBAC-cassette incorporated into the BacMam platform is a manifold tool offering advantages over standard transfection, in the scalability, efficiency and gene expression, which results in higher yields, shorter cultivation times and consequently cost-effective production processes (Reithofer et al., 20025).
Aims and methods.
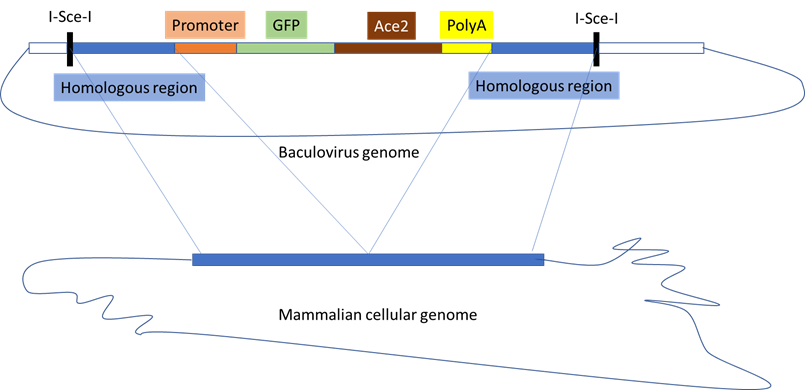
The aim of this study is to further develop and evaluate the potential of baculovirus-mediated transduction for transferring gene expression cassettes into various mammalian cell types and to achieve site-specific integration of target genes into the mammalian genome. The expression cassette encoded within the baculoviral genome is flanked by sequences homologous to the selected genomic integration locus, specifically the AAV safe-harbor site (Smith et al., 2008). In addition to this optimized expression cassette, designed for high-level recombinant protein production, the baculovirus vector also expresses a site-specific homing endonuclease (I-SceI), which introduces double-strand breaks adjacent to the homologous flanking regions. This cleavage event releases the expression cassette along with its homology arms from the baculoviral genome, thereby facilitating its integration into the designated genomic target site. The residual baculoviral genome becomes linearized and is subsequently degraded by cellular nucleases (Figure 1). Initially, several reporter constructs will be tested, including a gene encoding GFP fused to the SARS-CoV-2 receptor ACE2. Recombination frequency and clonal stability will be assessed, and resulting cell lines will be characterized for gene copy number and potential off-target integrations. Furthermore, the system will be applied to generate knockout cell lines and to evaluate the simultaneous integration of distinct target genes into separate genomic loci.
REFERENCES
Reithofer, M., Huber, S., Grabherr, R. (2025) Establishment of the REMBAC-cassette, a rapid, efficient and manifold BacMam tool for recombinant protein expression. J. Biotechnology 398 (12) DOI: 10.1016/j.jbiotec.2024.12.0118
Smith, J. R. et al. Robust, persistent transgene expression in human embryonic stem cells is achieved with AAVS1-targeted integration. Stem Cells 26, 496–504 (2008). https://doi.org/10.1634/stemcells.2007-0039
