Transglycosylation of Microbial β-N-Acetylhexosaminidases for the Biosynthesis of Human Milk Oligosaccharide Precursor Structure
SUPERVISOR: Thu-Ha Nguyen
Background.
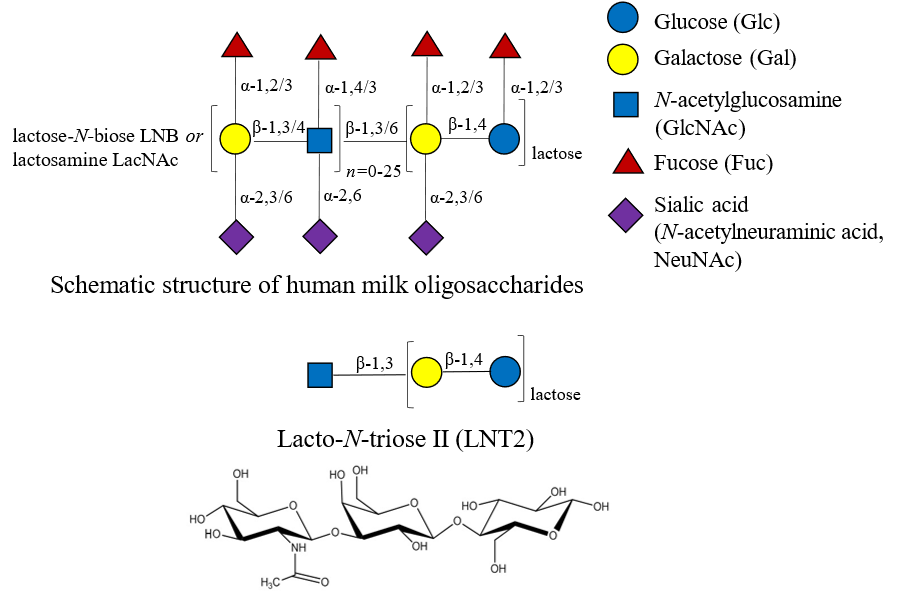
Human milk oligosaccharides (HMOs), constituting a substantial portion of human milk, serve as a primary source of prebiotics for infants (Selvamani et al., 2023). While HMOs exhibit promising potential for both infants and adults, their large-scale production is constrained by complex structures (Intanon et al., 2014). Efficient synthesis of homogeneous HMOs has gained significant attention due to their potential applications in human milk science and beyond. Among the synthetic routes, enzymatic synthesis using glycosidases is a promising approach and of great interest. β-N-Acetylhexosaminidases (EC 3.2.1.52) are among interesting candidates as they exhibit intrinsic transglycosylation capabilities, making them highly favorable for the biocatalytic synthesis of HMO precursor structure (Muschiol et al., 2020). Lacto-N-triose II (LNT2, GlcNAc-β1,3-Lactose) is a key HMO precursor structure formed by linking GlcNAc to β-lactose via a β(1→3) glycosidic bond. Elongation of LNT2 with galactose will result in lacto-N-neotetraose (LNnT, Gal-β1,4-GlcNAc-β1,3-Gal-β1,4-Glc) or lacto-N-tetraose (LNT, Gal-β1,3-GlcNAc-β1,3-Gal-β1,4-Glc), which are the backbone HMO structures. Some β-N-acetylhexosaminidases can catalyze the transfer of GlcNAc from the glycosyl donors such as pNP-GlcNAc and chitobiose (GlcNAc)2 to β-lactose to synthesize LNT2 (Zeuner et al., 2016). In this work, we will investigate novel β-N-acetylhexosaminidases and their biochemical properties and transglycosylation potentials for the biosynthesis of LNT2.
Aims and methods.
The proposed project aims: (i) to explore novel microbial β-N-acetylhexosaminidases based on phylogeny and sequence similarity networks; (ii) to study biochemical and structural properties of selected enzymes; (iii) to determine transglycosylation activities and the extent of N-acetyl-D-glucosamine (GlcNAc) transfer onto lactose of the selected enzymes; (iv) to identify potential residues involved in catalysis or in binding acceptor sugar (lactose), hence in transglycosylation; (v) to engineer enzyme variants with improved transglycosylation activities for enhanced yield of LNT2 formed.
This study will screen for novel β-N-acetylhexosaminidases based on phylogeny and sequence similarity networks. The genes encoding the selected enzymes will be cloned and overexpressed in Escherichia coli. The successfully overexpressed recombinant enzymes will be characterized pertaining to properties that are relevant to the application in biocatalytic transformations for the formation of LNT2. Structural models of the selected β‑N‑acetylhexosaminidases will be predicted by AlphaFold and visualized in PyMOL; molecular docking will be used to identify key catalytic residues involved in transglycosylation. Generation of variants based on r
ationalization of beneficial mutations in order to improve the formation of LNT2 will be performed.
REFERENCES
Selvamani, S., Kapoor, N., Ajmera, A., El Enshasy, H. A., Dailin, D. J., Sukmawati, D., ... & Abomoelak, B. (2023). Prebiotics in New-Born and Children’s Health. Microorganisms, 11(10), 2453. Intanon, M., Arreola, S. L., Pham, N. H., Kneifel, W., Haltrich, D., & Nguyen, T.-H. (2014). Nature and biosynthesis of galacto-oligosaccharides related to oligosaccharides in human breast milk. FEMS Microbiology Letters, 353(2), 89-97.
Muschiol, J., Vuillemin, M., Meyer, A. S., & Zeuner, B. (2020). β-N-Acetylhexosaminidases for carbohydrate synthesis via trans-glycosylation. Catalysts, 10(4), 365.
Zeuner, B., Nyffenegger, C., Mikkelsen, J. D., & Meyer, A. S. (2016). Thermostable β-galactosidases for the synthesis of human milk oligosaccharides. New Biotechnology, 33(3), 355–360.
