Biochemistry and Structure of Thyroid Peroxidase
SUPERVISOR: Vera PFANZAGL
Background.
The multidomain membrane protein Thyroid peroxidase (TPO) plays a key role in the biosynthesis of the thyroid hormones thyroxine (T4) and triiodothyronine (T3) [1]. TPO is nearly exclusively expressed in the thyroid gland and localized on the outer surface of the apical membranes of thyrocytes. There it catalyzes the hydrogen peroxide (H2O2)-dependent oxidation of iodide for iodination of tyrosine residues on the 660 kDa glycoprotein Thyroglobulin (TG) and the subsequent coupling reaction of iodinated tyrosine residues to form the thyroid hormones triiodothyronine (T3) and thyroxine (T4). As T4 and T3 are essential for growth, neuronal development, reproduction, and regulation of energy metabolism, thyroid hormone biosynthesis is a highly regulated process that is guided by the complex cellular organization of the thyroid gland follicle and involves the concerted action of multiple enzymes and proteins dedicated to H2O2 production or iodide transport and recycling.
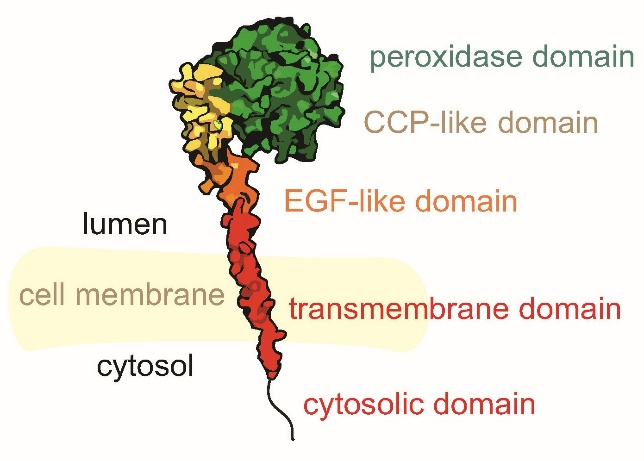
In contrast to the homologous and much better-studied soluble heme peroxidases MPO, EPO, and LPO, TPO is a multidomain transmembrane glycoprotein (Figure 1). The main enzymatically active domain is the peroxidase domain, which harbors the covalently linked heme cofactor. Additional domains include a transmembrane domain and several domains with unknown function. These are the complement control protein (CCP)-like domain, the epidermal growth factor (EGF)-like domain, and a large intracellular domain with no homologous scaffold. A recent low-resolution cryo-EM structure of the extracellular domains identified a cysteine bridge between the CCP-like and the peroxidase domain [2]. For the full-length protein, only computational models based on homologous dimeric MPO exist [3].
Aims & Methods.
To date, many open questions persist regarding both TPO catalysis and structure. Little is known about the structure-function relationship of TPO itself and how TPO catalyzes the phenolic coupling of iodotyrosines. In addition, TPO is at the core of two autoimmune thyroid diseases, Hashimoto’s and Graves’ disease, that combined afflict nearly 2% of the global population. The reason behind the high prevalence of TPO autoantibodies remains unclear. The only two clinically approved TPO inhibitors, targeting TPO hyperactivity in patients with Graves’ disease, are not specific and have poorly understood modes of action. A better understanding of TPO biochemistry and structure is clearly needed to address these open questions.
In this thesis, TPO oligomerization and activity will be probed both in vivo using the human thyroid cell line Nthy-Ori-3-1 and in vitro with recombinantly expressed TPO variants. The thesis will consist of two parts:
(I) To study the role of individual domains in TPO oligomerization, different TPO variants, lacking domains or with single mutations, will be fluorescently labeled and analyzed using single-molecule fluorescence microscopy (in cooperation with Mario Brameshuber, Technical University Vienna, Austria).
(II) To establish stable cell lines for recombinant production of TPO variants and optimize protein production and heme incorporation. Recombinant TPO variants will then be used for structure analysis by X-ray crystallography and cryo-EM as well as in solution using circular dichroism spectroscopy and HPLC analysis. Detailed biochemical characterization by time-resolved multi-mixing UV-vis stopped-flow studies will be performed to analyze the kinetics of substrate oxidation and the effect of known inhibitors. These studies will provide valuable insights into the biochemistry of hormone biosynthesis and the mechanism of action of known inhibitors of TPO.
REFERENCES
[1] Carvalho, DP and Dupuy C. (2017) Thyroid hormone biosynthesis and release. Molecular and Cellular Endocrinology, 458, 6-15, DOI: https://doi.org/10.1016/j.mce.2017.01.038
[2] Baker S, Miguel RN, Thomas D, Powell M, Furmaniak J, Smith BR. Cryo-electron microscopy structures of human thyroid peroxidase (TPO) in complex with TPO antibodies. J Mol Endocrinol. 2023 Jan 24;70(3):e220149. doi: https://doi.org/10.1530/JME-22-0149
[3] Le SN, Porebski BT, McCoey J, Fodor J, Riley B, Godlewska M, et al. (2015) Modelling of Thyroid Peroxidase Reveals Insights into Its Enzyme Function and Autoantigenicity. PLoS ONE 10(12): e0142615. DOI: https://doi.org/10.1371/journal.pone.0142615
