Probing the active site of pyranose dehydrogenase – covalent attachment of the FAD and vicinity of the isoalloxazine
SUPERVISOR: DIETMAR HALTRICH
Project assigned to: MICHAEL GRAF
Background.
Pyranose dehydrogenase (PDH, EC 1.1.99.29) is a member of the large family of FAD-containing sugar oxidoreductases and was only recently discovered. Its primary sequence shows significant similarity with several members of the GMC (glucose-methanol-choline) oxidoreductase family. It catalyses the oxidation of a wide range of carbohydrates, revealing a unique and amazingly varied catalytic versatility with respect to both the regioselectivity of the reaction and the substrate specificity. Depending on the source of the enzyme and the sugar substrate, (di)oxidation at C1, C2, C3, C1/2, C2/3 or C3/4 was described. In the ensuing oxidative half-reaction electrons are transferred to various electron acceptors including (substituted) quinones, or (complexed) metal ions. Oxygen is only a very poor electron acceptor, if active at all. As many of these oxidised sugars are not accessible by either chemical or even enzymatic means, PDH is a promising tool for introducing carbonyl functionality into various sugars which can then serve as synthons or building blocks for further synthesis reactions. PDH is furthermore of significant interest as a component in biofuel cells because of this relaxed specificity and activity with e.g. a number of sugars found as constituents of lignocellulose.
PDH is a monomeric, glycosylated flavoprotein of ∼65 kDa containing covalently bound FAD. A growing number of flavoenzymes have been reported to contain flavin covalently linked to a cysteine, histidine or tyrosine residue of the polypeptide chain. Yet, covalent attachment of flavin cofactors to enzymes is a relatively rare phenomenon (only approx. 10% of all flavoproteins are estimated to contain covalently bound flavin), and the number of different types of flavinylation observed is limited. Only recently, proteins containing bi-covalently attached FAD have been reported. The double-attachment flavinylation of the 6-S-cysteinyl, 8-α-N1-histidyl type has been shown for few members of the vanillyl alcohol oxidase family including gluco-oligosaccharide oxidase, berberine bridge enzyme, hexose oxidase, and chito-oligosaccharide oxidase through structural studies, mass spectrometry, and structural models. The exact reason why some enzymes tether a flavin by covalent attachment while others do not is not fully understood to date. Covalent flavinylation may serve to modulate the redox properties of an oxidoreductase, stabilise the correct conformation, or protect the coenzyme from modification and inactivation (Heuts et al., 2009). Based on a structural model we predicted a bi-covalent attachment of the FAD cofactor in PDH. This is the first example of this type of attachment in a member of the GMC family. Furthermore, we predicted a novel type of bi-covalent flavinylation with both an 8-α-N3-histidyl and a 9-S-cysteinyl linkage, which is unprecedented.
PDH has been purified and characterised in detail by our group using Agaricus xanthoderma and A. meleagris as the source of the enzyme (Kujawa et al., 2007, Sygmund et al. 2008, Kittl et al., 2008). Only recently we succeeded in overexpression of the pdh gene from A. meleagris in Aspergillus nidulans as well as in Pichia pastoris in levels of up to 200 mg recombinant protein per L, which paves the road to in-depth studies on PDH, e.g. on structure/function relationships. Furthermore, we obtained first diffracting crystals of the deglycosylated recombinant enzyme in collaboration with Prof. Divne, KTH Stockholm, and hence we are very optimistic to obtain the first structure of this enzyme in the near future.
Aims and methods.
This project is aiming at elucidating the role and importance of the bi-covalent attachment of FAD and of certain amino acid residues in the close vicinity of the isoalloxazine ring. To provide answers to these questions, we will mutate the residues involved in generation of the bi-covalent linkage (Cys99 and His102) as well as selected invariable residues of interest (His510 on the re face of the flavin near the C4a-N5 locus according to the structural model; His554 on the re face in proximity of the N1-C2=O2 locus). The latter position is chosen because it was shown that positively charged groups that are in contact with the isoalloxazine ring can be functionally relevant by stabilising the reduced cofactor and thus modulate the redox potential (Fraaije & Mattevi, 2000, Heuts et al., 2009). Further interesting amino acid positions will be identified through more detailed models in collaboration with OOSTENBRINK or once the structure is available.
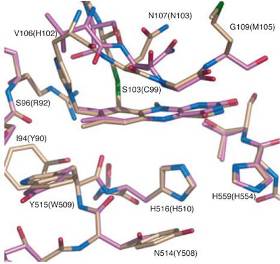
Proposed research work will include (i) elucidation of the spectral properties of wild-type PDH and its variants by using various spectrometric methods (UV-Vis, fluorescence, CD spectroscopy); (ii) steady-state kinetic analysis of wt and variant PDH; (iii) rapid kinetic studies to elucidate the effect of abolishing a single covalent linkage (C99A, C99S, H102A, H102N) as well as both linkages (C99A/H102A double mutant) on the reductive and oxidative half-reactions of PDH. Similarly, selected variants of His510 (H510A, H510N, H510R) and His554 (H554A, H554N, H554R) will be studied by stopped-flow spectrometry; (iv) redox potential determination of wild-type and mutant PDH by using the xanthine/xanthine oxidase method and/or an optically transparent thin-layer electrode (OTTLE) cell; (v) measurement of thermodynamic and kinetic stability of wt and variant PDH (determination of melting temperature and half-life of activity τ1/2) to assess the effects of covalent attachment on stability/unfolding; (vi) determination of the crystal structure (wt and variants) in collaboration with the structural biology laboratory at KTH (DIVNE).
Fraaije, M. W., Mattevi, M. (2000) Flavoenzymes: diverse catalysts with recurrent features. Trends Biochem. Sci. 25, 126–132
Heuts, D. P., Scrutton, N. S., McIntire, W. S., Fraaije, M. W. (2009) What's in a covalent bond? FEBS J. 276, 3405–3427
Kittl, R., Sygmund, C., Halada, P., Volc, J., Divne, C., Haltrich, D., Peterbauer, C. (2008) Molecular cloning of three pyranose dehydrogenase-encoding genes from Agaricus meleagris and analysis of their expression by real-time PCR. Curr. Genet. 53, 117–127
Kujawa, M., Volc, J., Halada, P., Sedmera, P., Divne, C., Leitner, C., Peterbauer, C., Haltrich, D. (2007) Properties of pyranose dehydrogenase purified from the litter-degrading fungus Agaricus xanthoderma. FEBS J. 274, 879–894
Sygmund, C., Kittl, R., Volc, J., Halada, P., Kubatova, E., Haltrich, D., Peterbauer, C. K. (2008) Characterization of pyranose dehydrogenase from Agaricus meleagris and its application in the C-2 specific conversion of D-galactose. J. Biotechnol. 133, 334–342
