Regulation of the expression of recombinant pentameric IgMs
SUPERVISOR: RENATE KUNERT
Project assigned to: VERONIKA CHROMIKOVA
Background.
Secreted proteins of complex structures are processed in the endoplasmic reticulum by different mechanisms influencing the structure and functionality of the mature molecule. In contrast to IgG molecules the highly complex IgM proteins are characterized by a heterodimeric μ-kappa/lambda heteropentamer that is linked by the joining (j-) chain. Besides the pentameric form, the second IgM variant found in plasma consists of a hexameric structure that is not connected via j-chain linkage and has unknown functions.
The scientific challenge is to define conditions enabling the release of functional oligomeric proteins into the culture supernatant. A further demand is to prevent aggregation and degradation of such complex molecules caused by the intrinsic structural and biophysical properties of these proteins. While plasma cells in their natural environment meet these demands properly; our recombinant CHO expression systems show controversial results. We demonstrated the expression of sufficient amounts of recombinant IgMs (Wolbank et al., 2003; Kunert et al., 2004) after a class switch from the respective IgGs. However, recombinant expression resulted in varying patterns of IgM with a large amount of low molecular weight isoforms. In case of IgGs the protein primary sequence influences the structural stability (Calarese et al., 2003) and therefore we conclude that a characteristic feature of naturally occurring IgMs (high homology to the germline with only minor somatic mutations) also plays an important role in proper IgM assembly. Additionally, the natural environment of plasma cells differs significantly from the protein-free media conditions in CHO bioreactors and therefore expression conditions must be evaluated carefully.
Based in these facts, this project is focused on the systematic investigation of regulatory and environmental factors influencing IgM assembly and secretion.
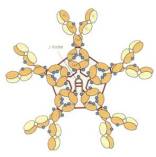 |
Aims and methods.
Parameters to investigate the influence on recombinant IgM expression will be (a) cell type and tissue selected for IgM expression, (b) environmental conditions necessary for induction/activation of endoplasmic reticulum and trans-Golgi network processing, and (c) protein engineering of IgM molecules to combine antigen specificity with the formation of pentameric structures.
In a first step vectors will be designed for recombinant expression of μ-, kappa- and joining- chains to generate different recombinant cell lines expressing IgM molecules with different specificity in different host cells. Therefore different IgMs will be expressed under strong regulatory elements in CHO cells and host cell lines of myeloid origin in serum-containing and protein-free media. Tasks to be performed will be the analysis of the secreted IgMs (bioactivity, oligomerization status) and the comparison of secreted and intracellularly accumulating IgMs.
Careful interpretation of the above mentioned analyses will culminate in the followed two scenarios:
(a) In case of host cell dependent proper expression, we will analyse the cell physiological differences of various host cells on a molecular level or will be able to define environmental requirements necessary for the expression of pentamers. This will be the basis for cellular engineering and product related definition of host cells for the intended purpose and cultivation conditions for complex proteins.
(b) If the primary amino acid sequence of the antibody by itself is the predominant factor influencing or dominating the generation of pentameric structures, a completely new approach for specific IgM production must be developed that is shortly described here:
Despite the fact that numerous antibodies have been crystallized in complex with their corresponding antigen, the impact of the respective variable region on the structure of the remainder of the antibody molecule is not well understood. It might be possible that the primary amino acid sequence of variable regions influences the formation of pentameric structures. Therefore our approach is to design modified variable antibody regions, which meet both demands, the antigen specificity of a distinct antibody and the structure of germ- line related antibodies.
Calarese, D. A., Scanlan, C. N., Zwick, M. B., Deechongkit, S., Mimura, Y., Kunert, R., Zhu, P., Wormald, M. R., Stanfield, R. L., Roux, K. H., Kelly, J. W., Rudd, P. M., Dwek, R. A., Katinger, H., Burton, D. R., Wilson, I. A. (2003) Antibody domain exchange is an immunological solution to carbohydrate cluster. Science 300, 2065-2071
Kunert, R., Wolbank, S., Stiegler, G., Weik, R., Katinger, H. (2004) Characterization of molecular features, antigen-binding, and in vitro properties of IgG and IgM variants of 4E10, an anti-HIV type 1 neutralizing monoclonal antibody. AIDS Res. Hum. Retrov. 20, 755-762
Wolbank, S., Kunert, R., Stiegler, G., Katinger, H. (2003) Characterization of human class-switched polymeric (immunoglobulin M [IgM] and IgA) anti-human immunodeficiency virus type 1 antibodies 2F5 and 2G12. J. Virol. 77, 4095-4103
