Engineering Pichia pastoris for antibody surface display
SUPERVISOR: MATTANOVICH DIETHARD
Project assigned to: LINA HEISTINGER
Background.
Antibody surface display is a technique used for the screening of large libraries of antibodies for their affinity to a target antigen. To be able to obtain antibodies with the required characteristics, the screening library used requires a large size and high variability. In the yeast Saccharomyces cerevisiae, an increase in antibody diversity is achieved on one hand by site directed mutagenesis employing in vivo recombination, and on the other hand by expressing the antibodies’ light and heavy chains in different haploid strains which are then mated to bring the different chains together. The main disadvantage of S. cerevisiae is the low product yield once a soluble antibody should be produced. Therefore, a change in host system is often required for the production of the desired antibody for further testing.
The yeast Pichia pastoris was shown to be well suited for the expression of antibodies. However, due to certain cellular characteristics, P. pastoris is not as well suited for the establishment of antibody libraries. Within this project, P. pastoris strains will be engineered to be more suitable for the generation and screening of IgG surface display libraries.
Aims and methods.
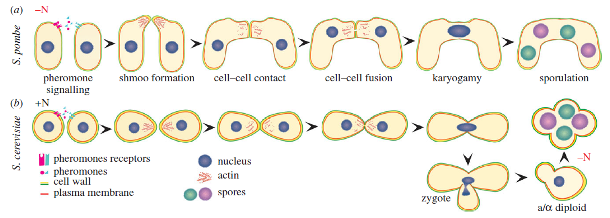
The aim of the project is to develop P. pastoris strains for surface display and screening of IgG libraries. The first step in engineering P. pastoris will be the improvement of transformation efficiency. This should be achieved by increasing the frequency of homologous recombination of DNA fragments into the genome. At the same time the length of the homologous ends required for recombination should be decreased.
The second part of the project consists of the establishment of a mating strategy for P. pastoris to increase library diversity. It has been shown that the variability of antibody libraries can be greatly increased by mating a yeast library producing antibody's heavy chains with a second library producing only light chains. However, in contrast to S. cerevisiae laboratory strains, P. pastoris strains do not differ explicitly in mating type and, once mated, do not maintain a diploid state. Manipulation of the genes responsible for the switching of mating type will be the main strategy to convert P. pastoris into a heterothallic yeast with a determined mating type. The obtained strains will be tested for their mating efficiency. Furthermore, different approaches to maintain the mated cells in a diploid state will be investigated.
Merlini, L., Dudin, O., Martin, S.G. (2013) Mate and fuse: how yeast cells do it. Open Biol 3: 130008.
