Efficient free energy and enhanced sampling calculations of protein-protein interactions
SUPERVISOR: CHRIS OOSTENBRINK
Project assigned to: ZUZANA JANDOVA
Background.
Modern molecular life sciences place a large emphasis on the complex interactions between proteins and corresponding networks. In the pharmaceutical sciences, the focus is shifted from small-molecule drugs to so-called ‘biologicals’, which may be complex protein systems. Computational descriptions of such interactions lead to insight at the molecular level and predictions of affinities between proteins open the way to the rational design of novel therapeutics.
In this project, members of the 14-3-3 protein family and a typical binding partner are chosen as model proteins to apply various novel methods. The 14-3-3 proteins combine interesting physicochemical features with high biological/medicinal relevance. 14-3-3 proteins (14-3-3s) are important modulators of several key signaling proteins regulating critical biological activities such as cell cycle control, cell growth, proliferation, and apoptosis (1,2,3). 14-3-3s are associated with oncogenic proteins, such as Raf-1, Bcr-Abl, Bcr, and polyoma middle T-antigen, and are particularly abundant in the mammalian brain where they have been associated to the pathophysiology of various neurological and neurodegenerative disorders, including Creutzfeldt-Jakob, Alzheimer’s, Parkinson’s and polyglutamine repeat diseases (4,5,6).
Aims and methods.
An accurate description of protein-protein interactions and the relevant free energy differences crucially depend on appropriate sampling of all relevant conformational states, both in the bound as well as in the unbound state of the binding partners. While relatively efficient computational tools have been described for the interactions between proteins and small molecules, the protein-protein interactions pose additional challenges due to the large diversity in amino acid sequences and the intrinsic flexibility of protein structures.
In the current proposal, we will develop efficient free energy methods and enhanced sampling tools to compute the binding free energy for phosphylated peptides binding to the 14-3-3 protein.
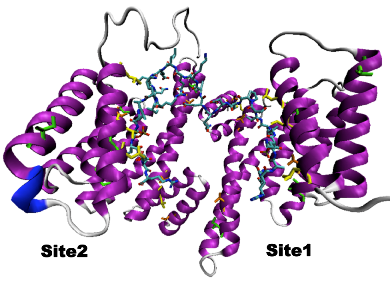
Crystal structures of various 14-3-3 proteins are available and show that each monomer is composed of nine α-helices arranged in antiparallel fashion to form an amphipathic groove that mediates the bindings of the partners in a serine/threonine-phosphorylation dependent way. The binding process will be described using Hamiltonian replica exchange (H-REMD) calculations, in combination with distance field distance restraints (7,8). We will apply different schemes of H-REMD where enhanced sampling results from the application of distance restraints at distances of increasing lengths. The main advantage of applying H-REMD over sequential umbrella sampling simulations comes from the fact that the replicas exchange in a controlled manner exploiting natural movements rather than trying to overcome steric obstacles on the ligand pathway. Moreover, multiple binding paths can be sampled reversibly, where the application of distance-field restraints will ensure that the peptide finds the proper position in the 14-3-3 binding groove.
Overall, the developed methods will be applicable for a wide variety of protein-protein interactions and the enhanced sampling tools will allow for the calculation of complex potential of mean force profiles to describe the interaction between very flexible molecules.
1. Morrison, D. K. The 14-3-3 proteins: integrators of diverse signaling cues that impact cell fate and cancer development. Trends Cell Biol. 2009, 19, 16-23.
2. Gardino, A. K. Structural determinants of 14-3-3 binding specificities and regulation of subcellular localization of 14-3-3- ligand complexes: A comparison of the X-ray crystal structures of all human 14 3 3 isoforms. Semin. Canc. Biol. 2006, 16, 173-182.
3. Freeman, A. K., Morrison, D. K. 14-3-3 Proteins: diverse functions in cell proliferation and cancer progression. Semin. Cell Dev. Biol. 2011, 22, 681-687.
4. Steinacker, P., Aitken, A., Otto, M., 14-3-3 proteins in neurodegeneration. Semin. Cell Dev. Biol. 2011, 22, 696-704.
5. Mackie, S., Aitken, A., Novel brain 14-3-3 interacting proteins involved in neurodegenerative disease. FEBS J. 2005, 272, 4202-4210.
6. Shimada, T., Fournier, A. E., Yamagata, K. Neuroprotective Function of 14-3-3 Proteins in Neurodegeneration. BioMed. Res. Int. 2013, 2013, 564534.
7. Hritz, J., Oostenbrink, C. Optimization of replica exchange molecular dynamics by fast mimicking. Journal of Chemical Physics 2007, 127, 204104.
8. de Ruiter, A., Oostenbrink, C. Protein-ligand binding from distancefield distances and Hamiltonian replica exchange simulations. Journal of Chemical Theory and Computation 2013, 9, 883-892.
