Oral health: Development of genetic tools for Selenomonas sputigena to unravel mechanisms of dental plaque colonization
SUPERVISOR: CHRISTINA SCHÄFFER
Background.
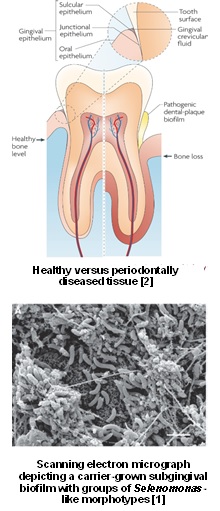 The oral cavity provides a habitat for about 700 prokaryote species, which form polymicrobial biofilms on oral tissues and teeth [3]. These highly specialized microbial communities de-velop through precise spatial and temporal interactions; dis-ruption of homeostasis between the different species can cause a shift from oral commensal to pathogenic bacteria lead-ing to the onset of periodontitis, a multifactorial inflammatory disease [2]. While the so called "red complex" bacteria Tannerella forsythia, Porphyromonas gingivalis and Treponema denticola have been identified as the causative agents for perio-dontitis [4], the contribution of the numerous other bacteria present in dental plaque biofilms to disease development are poorly understood.
The oral cavity provides a habitat for about 700 prokaryote species, which form polymicrobial biofilms on oral tissues and teeth [3]. These highly specialized microbial communities de-velop through precise spatial and temporal interactions; dis-ruption of homeostasis between the different species can cause a shift from oral commensal to pathogenic bacteria lead-ing to the onset of periodontitis, a multifactorial inflammatory disease [2]. While the so called "red complex" bacteria Tannerella forsythia, Porphyromonas gingivalis and Treponema denticola have been identified as the causative agents for perio-dontitis [4], the contribution of the numerous other bacteria present in dental plaque biofilms to disease development are poorly understood.
Physical interactions within biofilms are facilitated by special-ized cell envelope structures, such as glycoconjugates, with the cell surface glycobiology of human oral bacteria playing an important role in plaque formation, inter-species communica-tion and virulence, as has been shown for the glycosylated S-layer proteins TfsA and TfsB in T. forsythia [5].
The Gram-negative anaerobe Selenomonas sputigena is frequently co-isolated with T. forsythia and occurs in elevated levels in patients with generalized aggressive periodontitis [6]. The rich glycoproteome of S. sputigena suggests, that this oral species employs various glycobiology-based strategies for oral colonization and survival. Putative glycosylation of its flagella and the prediction of uncommon sugar residues, such as L-fucose and pseudaminic acid make it an interesting candidate to study glycobiology aspects in the context of periodontal disease and biofilm architecture.
Aims and methods.
In order to further investigate the glycobiology of S. sputigena, the primary aim of this project is to develop genetic tools for the manipulation of this microorganism. Targeted knock-outs of glycan biosynthesis and glycosylation active enzymes will allow us to generate mutants with defined cell envelope defects. Through the modification of the glycosylation of key glycoconjugates we will be able to assess their function with regard to biofilm formation and community interactions, as well as potential pathogenicity mechanisms.
1.Drescher, J., et al., Molecular epidemiology and spatial distribution of Selenomonas spp. in subgingival biofilms. Eur J Oral Sci, 2010. 118(5): p. 466-74.
2.Darveau, R.P., Periodontitis: a polymicrobial disruption of host homeostasis. Nat Rev Microbiol, 2010. 8(7): p. 481-90.
3.Paster, B.J., et al., The breadth of bacterial diversity in the human periodontal pocket and other oral sites. Periodontol 2000, 2006. 42: p. 80-7.
4.Socransky, S.S., et al., Microbial complexes in subgingival plaque. J Clin Periodontol, 1998. 25(2): p. 134-44.
5.Posch, G., et al., Glycobiology Aspects of the periodontal pathogen Tannerella forsythia. Biomolecules, 2012. 2(4): p. 467-82.
6.Goncalves, L.F., et al., Levels of Selenomonas species in generalized aggressive periodontitis. J Periodontal Res, 2012. 47(6): p. 711-8.
