Plant glyco-engineering towards synthesis of recombinant proteins with polysialylated N-glycans
SUPERVISOR: HERTA STEINKELLNER
Project assigned to: SOMANATH KALLOLIMATH
Background.
Plants have been proposed as bioreactors for recombinant protein production because they can generate large volumes of proteins at low cost (Castilho and Steinkellner 2012). Plants can transiently express target proteins within 1 to 2 weeks (Gleba et al., 2012) and importantly they are capable of performing similar post-translational modifications like mammals, particularly with respect to glycosylation. Concerns with plant specific N-glycosylation have been addressed extensively and over the past years we and others have concentrated efforts on modulating plant N-glycosylation towards the production of recombinant proteins with human-like structures (Castilho and Steinkellner, 2012).
An important factor for the clinical and commercial success of protein based drugs lies in optimal bioavailability, which is a major challenge for pharmaceutical product development. Polysialylated structures (PSA) have the ability to modulate pharmacokinetics, bio-distribution, and bioavailability of recombinant proteins. These large negatively charged and highly hydrated chains are naturally present on neural cell adhesion molecules (NCAMs) in humans and act as a key regulator of cell surface interactions. Notably, attachment of PSA to recombinant proteins has been shown to increase their bioavailability (Chen et al., 2012; Ilyushin, et al. 2012). Due to their enormous complexity it is difficult to engineer this structure in vitro and in vivo.
Aims and methods.
We aim to reconstruct PSA on various pharmaceutically relevant recombinant proteins (e.g. erythropoietin, butyrylcholinesterase, antibody fragments, NCAM) in plants. In humans, two Golgi polysialyltransferases, ST8SiaII (STX) and ST8SiaIV (PST), are known to carry out polysialylation. Both enzymes are able to synthesize long PSA chains in the presence of the donor substrate CMP-SiA. We will build on our recent results in in planta sialylation, where we demonstrated the synthesis of (multi-) sialylated structures on various proteins (Castilho et al., 2013).
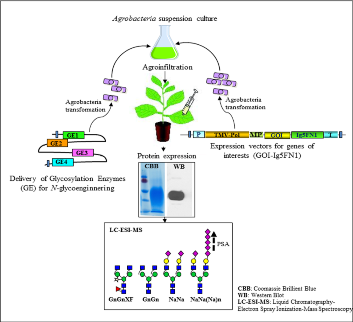
- 1. Transient expression and characterisation of the two human ST8Sia genes in N. benthaminana
- 2. Expression of Ig5FN1, the naturally polysialylated domain of NCAM (Hildebrandt et al., 2010)
- 3. Generation of therapeutically interesting proteins fused to Ig5FN1domain
-
4. Modulation of glycosylation will be achieved by co-expressing the reporter proteins (i) with mammalian genes necessary for protein α 2,6-sialylation (6 genes, Castilho et al., 2010) and (ii) polysialylation will be attempted by additionally expressing human polysialyltransferases ST8siaII and/or ST8siaIV
- 5. Purified fusion proteins will be analysed by liquid-chromatography electrospray ionization-mass spectrometry, to determine their N-glycosylation profile and evaluate the extent of our attempts on glycoengineering towards polysialylation (in cooperation with Friedrich Altmann)
- 6. PSA-engineered proteins will be subjected to different bioavailability assays, e.g. in mouse pharmacokinetic and bio-distribution studies (e.g. cell uptake). In addition engineered PSA on recombinant proteins provide a new tool for basic studies as the multiple function of this complex glycan formation is poorly investigated (Hildebrandt et al., 2010).
Castilho, A., Strasser, R., Stadlmann, J., Grass, J., et al., In planta protein sialylation through overexpression of the respective mammalian pathway. J. Biol. Chem. 2010, 285, 15923-15930.
Castilho, A., Steinkellner, H., Glyco-engineering in plants to produce human-like N-glycan structures. Biotechnol. J. 2012, 7, 1088-1098.
Castilho, A., Neumann, L., Gattinger, P., Strasser, R., et al., Generation of biologically active multi-sialylated recombinant human EPOFc in plants. PloS One 2013, 8, e54836.
Chen C, Constantinou A, Chester KA, Vyas B, Canis K, Haslam SM, et al.,. Glycoengineering approach to half-life extension of recombinant biotherapeutics. Bioconjug Chem. 2012; 23:1524-33.
Gleba YY, Tusé D, Giritch A. Plant Viral Vectors for Delivery by Agrobacterium. Curr Top Microbiol Immunol. 2013 (in press)
Hildebrandt H, Muhlenhoff M, Gerardy-Schahn R. Polysialylation of NCAM. Adv Exp Med Biol 2010, 663, 95-109
Ilyushin DG, Smirnov IV, Belogurov AA Jr, Dyachenko IA, Zharmukhamedova TI, et al., Chemical polysialylation of human recombinant butyrylcholinesterase delivers a long-acting bioscavenger for nerve agents in vivo. Proc Natl Acad Sci U S A. 2013, 110:1243-8. doi: 10.1073/pnas
