MAPK and JAK/STAT Signaling in Immune Regulation by Dendritic Cells
SUPERVISOR: LUKAS MACH
Project assigned to: KLARA SOUKUP
Background.
Dendritic cells (DCs) represent important communicators at the interface of the innate and the adaptive immune system. Their potential of inducing a potent and specific immune response by interacting with and activating primarily T cells makes them an attractive target for immunotherapy. In recent years, cancer immunotherapy based on the application of ex vivo activated DCs has emerged as a promising strategy to establish a potent and sustained anti-tumor immune response (e.g. Melief, 2008). Up to date, several early-phase clinical trials have demonstrated the safety and feasibility of this approach, and in 2010, the first therapeutic cancer vaccine for the treatment of metastatic prostate cancer was approved by the FDA. However, current approaches of DC-based cancer immunotherapy (DC-CIT) are struggling with high inter-individual variability of therapeutic efficacy, revealing the need for improving treatment strategies.
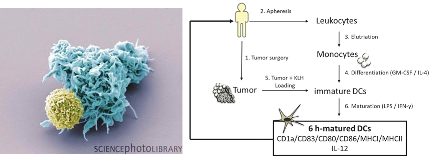
Besides the notion that different DC subsets may account for diverging immunological functions of these professional antigen-presenting cells (Palucka et al., 2010), previous studies have shown that DCs are capable of switching from a pro- towards an anti-inflammatory phenotype following stimulation by microbial danger signals (Luger et al., 2013). This functional change is characterized by a decrease in interleukin (IL)-12 secretion along with an upregulation of IL-10 production, which severely affects helper T cell polarization (Langenkamp et al., 2000). Such effects contribute to the attenuation of a generated anti-tumor immune response and are supposed to act as negative feedback mechanisms in order to prevent damage caused by excessive inflammatory activity.
Preliminary genome expression analyses revealed several potential candidates involved in directing DCs into the regulatory phenotype – among them distinct members of the mitogen-activated protein kinase (MAPK) and Janus kinase/signal transducer and activator of transcription (JAK/STAT) signaling pathways. Clarification of the role these molecules play in DCs will be crucial for the evaluation of their potential as promising candidates for DC-CIT efficacy improvement.
Aims and methods.
In this thesis project, the involvement of two specific signaling molecules in regulatory DC functions will be assessed - one of which forms part of the MAPK signal transduction pathway, whereas the other represents an important JAK/STAT signaling mediator.
In vitro, murine knock-out DCs will be used for functional assays comparing stimulatory capacities to wildtype DCs in the context of effector and helper T cell activation and differentiation. Also small molecule inhibitors will be used to explore the potential of applying specific kinase inhibitors in combination with DC-CIT in the future.
For investigating the physiological relevance of in vitro findings, conditional knock-out mouse models will be employed to study in vivo effects of DCs lacking activity of the signaling molecules in question. Furthermore, murine transplantable tumor models will be used to mimic clinical application of DC-CIT and assess the effects of applying candidate molecule-deficient DCs regarding reduction of tumor growth. In addition to already available syngeneic heterotopic tumor models, orthotopic models will be established for enhanced physiological authenticity. In order to investigate immunostimulatory and –regulatory effects mediated by wildtype and modified DCs, tumor tissue and peripheral organs (spleens, lymph nodes and peripheral blood) will be monitored for infiltration by inflammatory and regulatory cell types. For this purpose, flow cytometry as well as immunofluorescence staining approaches will be pursued.
Langenkamp, A., Messi, M., Lanzavecchia, A., and Sallusto, F. (2000) Kinetics of dendritic
cell activation: impact on priming of TH1, TH2 and nonpolarized T cells. Nat
Immunol 1(4), 311-316
Luger, R., Valookaran, S., Knapp, N., Vizzardelli, C., Dohnal, AM, Felzmann, T. (2013) Toll-like receptor 4 engagement drives differentiation of human and murine dendritic cells from a pro- into an anti-inflammatory mode. PLoS One 8(2), e54879
Melief CJM (2008) Cancer immunotherapy by dendritic cells. Immunity 29(3), 372–383
Palucka, K., Ueno, H., Zurawski, G., Fay, J., Banchereau, J. (2010) Building on dendritic cell subsets to improve cancer vaccines. Curr Opin Immunol 22(2), 258–263
