Engineering the interface of Fab fragments
SUPERVISOR: CHRISTIAN OBINGER
Project assigned to: SYLVIA DIETRICH
Background.
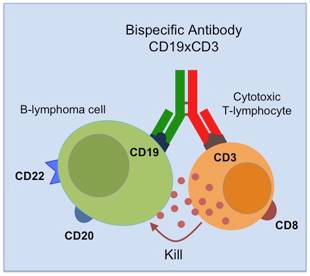
Monoclonal antibodies, the most frequently used immunotherapeutics to date, can combat pathogens by several mechanisms like mere binding (neutralization) or opsonisation for subsequent immune mechanisms, such as the complement system (complement dependent cytotoxicity, CDC) or cytotoxic effector cells (antibody-dependent cellular cytotoxicity, ADCC). Bispecific antibodies offer an elegant approach to improve antibody therapy by simultaneously binding two different targets by a single molecule. They can for instance directly cross-link a target cell with an effector cell that lacks the appropriate Fc receptor. They are thus able to recruit even cell subsets that would normally not participate in an immune response. Other possible therapeutic applications of bispecific antibodies are redirecting potential effector systems (molecules like enzymes, drugs, antigens, toxins etc; and cells like CTL, macrophages, natural killer cells, neutrophils, etc) to diseased areas and/or increasing neutralizing or stimulating activities of antibodies (Dübel, 2007).
Research and development in the antibody engineering field brought about new approaches for the generation of novel bispecific antibody formats and revived the interest in these molecules. Numerous methods for generating bispecific binding of the complete, Ig-like antibodies have been developed, e.g. knobs-into-holes (KiH) and SEED technology, both based on specific pairing of interface amino acids in the CH3 domain of heavy chains (Merchant et al. 1998, Davis et al. 2010). The main problem of these approaches for the production of bispecific antibodies is that the pairing of the light chains with the heavy chains in the Fab part can still, to a certain degree, occur at random, resulting in heterogeneous products secreted by the expressing cells, which make further downstream processing cumbersome. In conclusion, there are two problems to be solved for the successful production of a bispecific antibody: (i) effective induction of heterodimerization of the two heavy chains (KiH, SEED) and (ii) discrimination between the two light chain/heavy chain interactions (Schaefer et al., 2011). In the Christian Doppler Laboratory for Antibody Engineering (Department of Biotechnology, in partnership with Merck KGaA), we focus on solving the latter problem by using molecular biology tools and engineering the interface in Fab fragments.
Aims and methods.
The main purpose of the present project is to devise a strategy that allows specific and controlled pairing between heavy and light chains in the Fab region of heterodimeric antibody constructs. More specifically, mutations are introduced into the interface between VH and VL domains on one hand, and the CH1/CL interface on the other hand, that allow selective pairing with its respective counterpart, but not with the corresponding wild type or otherwise engineered chain. A generic solution that can be applied to different antibodies is preferred over a solution that needs case by case engineering.
Based on crystal structure analysis of several similar Fabs containing kappa light chain (available on the PDB) and a closer look at the possible interface amino acid interteractions, suitable candidates for mutagenesis are chosen. The mutagenesis is performed either by site directed mutagenesis of single amino acids or using a library approach, with which several mutations will be introduced simultaneously. The mutagenesis and first rounds of screening, selection and sorting of mutants are performed on Fabs displayed on the surface of Saccharomyces cerevisiae yeast cells. Both, light and heavy chains in the Fab molecule are fused to C-terminal peptide tags (e.g. His-Tag, myc-tag, FLAG-tag). These tags allow us to use fluorescent dye-conjugated antibodies for the detection in Fluorescence-activated cell sorting (FACS) and for purification of soluble Fab molecules in the later stages of the project. Interesting candidates will be expressed and purified from soluble fractions using Pichia pastoris or mammalian cell expression systems and characterized for antigen binding (ELISA, Octet) and for biophysical properties (SEC, DSC, CD, IEF). Putative final mutations will be introduced in other Fab molecules and /or SEED format for proof of concept and possible application in bispecific antibody engineering.
Dübel, S. (Ed.). (2007). Handbook of Therapeutic Antibodies. Weinheim, Germany: Wiley-VCH Verlag GmbH.
Schaefer, W., Regula, J. T., Bahner, M., Schanzer, J., Croasdale, R., Durr, H., Gassner, C., et al. (2011). Immunoglobulin domain crossover as a generic approach for the production of bispecific IgG antibodies. Proceedings of the National Academy of Sciences, 108(27).
Davis, J. H., Aperlo, C., Li, Y., Kurosawa, E., Lan, Y., Lo, K.-M., Huston, J. S. (2010). SEEDbodies: fusion proteins based on strand-exchange engineered domain (SEED) CH3 heterodimers in an Fc analogue platform for asymmetric binders or immunofusions and bispecific antibodies. Protein engineering, design & selection: PEDS, 23(4), 195-202.
Merchant, A. M., Zhu, Z., Yuan, J. Q., Goddard, A., Adams, C. W., Presta, L. G., Carter, P. (1998). An efficient route to human bispecific IgG. Nature biotechnology, 16(7), 677-81.
