Structural and functional characterization of Fcab-antigen interaction
SUPERVISOR: CHRISTIAN OBINGER
Project assigned to: ELISABETH LOBNER
Background.
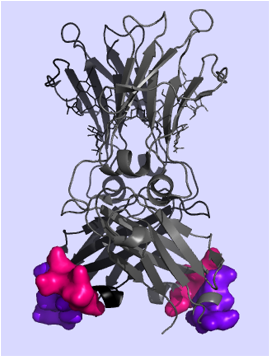
Monoclonal antibodies as therapeutic agents offer enormous potential in the treatment of various serious diseases such as autoimmune diseases, infectious diseases and cancer. Advantages of antibodies include high specifity, low off-target effects, high stability in serum and the capability to induce fragment-crystallizable (Fc)-mediated effector functions. The disadvantage of poor tissue penetration and intricate production can be managed by using fragments instead of a full-size antibody such as the Fc domain. The Fc domain comprises all effector functions for the communication with the immune system and thus offers an ideal format for engineering. New antibody formats – based on the Fc domain – have been developed by Christian Doppler Laboratory for Antibody Engineering that was found jointly with F–star. By using Modular Antibody Technology, novel binding sites were introduced to the C-terminal structural loops of the CH3 domains of the Fc, resulting in an Fcab (Fc antigen binding) (Wozniak–Knopp et al., 2010).
In previous projects Fcabs were generated by randomizing the AB and EF loops of the C-terminal tip of the CH3 domain in order to bind to the antigen Her2/neu that is a member of the epidermal growth factor receptor family. Overexpression of Her2/neu is observed in 20–25% of invasive breast cancer and is associated with a high aggressive phenotype and poor prognosis [Wong and Lee, 2012]. Wozniak-Knopp et al. produced the Fcab clone H10–03–6 by library sorting for binders against Her2/neu using FACS (fluorescence activated cell sorting). Traxlmayr et al. generated Fcabs (STAB1 and STAB19) with improved stability and affinity to Her2/neu, based on loop mutations of the H10-03-6.
 |
 |
Aims and methods.
The aim of the project is to investigate the structural and thermodynamic basis of Fcab-antigen interaction(s). In the beginning three different Fcabs (H10-03-6, STAB1, STAB19) will be tested for their interaction with the extracellular domain of Her2/neu by a broad set of biochemical and biophysical techniques. A necessary prerequisite is the establishment of recombinant production of all proteins in HEK 293 (human embryonic kidney) cells, followed by protein purification and quality control using SEC (size exclusion chromatography), DSC (differential scanning calorimetry) and MALLS (multi angle laser light scattering).
X–ray crystallography of Fcab structures will give an understanding of the impact of the different loop designs on loop conformation and intra– and interchain loop interactions. In addition, evaluation of the obtained structures by MD (molecular dynamics)–simulations will give further insight into the binding mode(s) of these novel antibody fragments with their target proteins.
To get a full picture of the interaction, MALLS and ITC (isothermal titration calorimetry) will be established. Thereby, important thermodynamic data will be obtained that allow elucidation of the enthalpic and entropic driving forces involved in the respective Fcab-antigen interaction. Additionally, information about binding stoichiometry and strength of binding under various conditions can be investigated.
Furthermore, in order to understand similarities and differences between crystal structure(s) and interaction in solution, epitope mapping through yeast display of the single Her2/neu domains will be set up. By elucidation of contact loops and residues, it is possible to obtain knowledge of the Fcab-antigen binding interface in solution. Together with data from co-crystallization, MD-simulations, MALLS and ITC, this will allow detailed analysis of Fcab–antigen interplay.
In addition to X–ray crystallography, we are also aiming at solving an Fcab structure in solution by NMR (nuclear magnet resonance). Therefore, isotopic labelling strategies (15N, 13C) of HEK produced Fcabs have to be established. Furthermore, the interaction of Fcab with the Her2/neu binding domain can be studied by NMR.
The project will be performed within the Christian Doppler Laboratory for Antibody Engineering. Cooperation partners include Chris Oostenbrink at BOKU (Institute for Molecular Modeling and Simulations)and the Department for Structural and Computational Biology at MFPL (University of Vienna).
Wozniak-Knopp, G., Bartl, S., Bauer, A., Mostageer, M., Woisetschläger, M., Antes, B., Ettl, K., Kainer, M., Weberhofer, G., Wiederkum, S., Himmler, G., Mudde, G. C., Rüker, F.(2010) Introducing antigen-binding sites in structural loops of immunoglobulin constant domains: Fc fragments with engineered HER2/neu-binding sites and antibody properties. Protein Eng. Des. Sel., 23 (4), 2010:289-297. doi: 10.1093/protein/gzq005.
Wong, A.L.A., Lee, SC. (2012) Mechanisms of resistance to trastuzumab and novel therapeutic strategies in Her2-positive breast cancer. Int. J. Breast Cancer, doi: 10.1155/2012/415170.
Traxlmayr, M.W., Lobner, E., Antes, B., Kainer, M., Wiederkum, S., Hasenhindl, C., Stadlmayr, G., Rüker, F., Woisetschläger, M., Moulder K., Obinger, C. (2012) Directed evolution of Her2/neu-binding IgG1-Fc for improved stability and resistence to aggregation by using yeast surface display. Protein Eng. Des. Sel., 26(4): 255-265. doi: 10.1093/protein/gzs102
