Antibody production in seed tissues: evaluating the subcellular deposition, N-glycan modification and functionality
SUPERVISOR: EVA STÖGER
Project assigned to: STANISLAV MELNIK
Background.
Plants are being developed as alternative large-scale production systems for recombinant therapeutic proteins because of their low costs and scalability. Among the many plant-based production systems that have been developed for pharmaceutical proteins, seeds have the useful advantage of accumulating proteins in a relatively small volume and in a protected environment. Several seed crops, including cereals, grain legumes and oilseeds, have been explored as production platforms. Maize is favoured particularly because of its short generation interval and high protein content, and a precedent has been established due to the use of maize for the commercial production of several protein technical reagents (for review see Stoger et al., 2005).
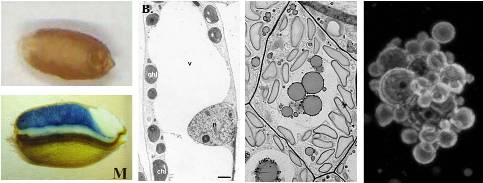
However, seed-based production frequently leads to different N-glycan structures and altered subcellular localization of recombinant proteins (Arcalis et al., 2004; van Droogenbroek et al., 2007; Abranches et al., 2008). Along these lines we have compared the subcellular deposition and N-glycosylation of a recombinant model glycoprotein in rice seeds and leaves, concluding that the storage function of rice endosperm may alter the fate of the recombinant protein (Drakakaki et al., 2006). With a seed-derived IgG1 from maize, an even more drastic trimming of glycan structures was observed, again in the context of protein deposition within storage organelles (Rademacher et al., 2008). It is not clear whether such behaviour is specific to the highly specialized cereal endosperm, or whether it occurs in other storage tissues as well. To address this question, we propose to study the deposition and modification of the broadly neutralizing human anti-HIV antibody 2G12 in embryo storage tissue of maize.
Aims and methods.
One aim of the project is to determine whether the observed tendency towards intracellular deposition of recombinant proteins in cereal endosperm is a consequence of the high functional specialization of storage cells in general, or an endosperm-specific attribute. We will evaluate whether glycan-modifying enzymes such as endo-N-acetylglucosaminidases are present and able to act on recombinant proteins targeted to the endomembrane system in non-endospermous parts of the cereal seed. Finally, seed-derived antibody glycoforms will be investigated for their functional performance, especially for Fc receptor binding and antibody-dependent cellular cytotoxicity (ADCC).
Genes encoding the heavy and the light chain of anti-HIV antibody 2G12 will be expressed in maize under the control of the embryo-specific rice globulin promoter. Control plants producing the same antibody with or without ER-retention signal in an endosperm-specific manner are readily available (Rademacher et al., 2008; Ramessar et al., 2008). In those plants, the antibody was mainly deposited within zein bodies and most of the glycans were trimmed extensively.
The antibody will be localized within the embryo using confocal and electron microscopy combined with immunofluorescence and immunogold detection of the heavy and light chain. The antibody will be purified from homozygous seeds using protein A affinity chromatography. Yield and efficiency of the purification will be compared to the endosperm-based production system, and the specific antigen-binding function of the purified antibody will be verified by ELISA and surface plasmon resonance analysis. N-glycan structures will be analysed by LC-ESI-MS as described (Rademacher et al., 2008). In vitro cell assays will be used to compare the HIV-neutralizing properties of the maize-produced antibodies and their CHO-derived counterpart. Most importantly, the seed-derived glycoforms of the antibody will be tested for their ability to bind to Fcγ receptors and to trigger antibody-dependent cell-mediated cytotoxicity (ADCC).
Abranches, R., Arcalis, E., Marcel, S., Altmann, F., Ribeiro-Pedro, M., Rodriguez, J., Stöger, E. (2008) Functional specialization of Medicago truncatula leaves and seeds does not affect the subcellular localization of a recombinant protein. Planta 227, 649-658
Arcalis, E., Marcel, S., Altmann, F., Kolarich, D., Drakakaki, G., Fischer, R., Christou, P., Stöger, E. (2004) Unexpected deposition patterns of recombinant proteins in post-ER compartments of wheat endosperm. Plant Physiol. 136: 3457-3466
Drakakaki, G., Marcel, S., Arcalis, E., Altmann, F., Gonzalez-Melendi, P., Fischer, R., Christou, P., Stöger, E. (2006) The intracellular fate of a recombinant protein is tissue-dependent. Plant Physiol. 141:578-586
Rademacher, T., Sack, M., Arcalis, E., Stadlmann, J., Balzer, S., Altmann, F., Quendler, H., Stiegler, G., Kunert, R., Fischer, R. Stöger, E. (2008) Recombinant antibody 2G12 produced in maize endosperm efficiently neutralizes HIV-1 and contains predominantly single-GlcNAc N-glycans. Plant Biotechnol. J. 6, 189-201
Ramessar, K., Rademacher, T., Sack, M., Stadlmann, J., Platis, D., Stiegler, G., Labrou, N., Altmann, F., Ma, J., Stöger, E., Capell, T., Christou, P. (2008) Cost-effective production of a vaginal protein microbicide to prevent HIV transmission. Proc. Natl. Acad. Sci. U S A. 105, 3727-3732
Stöger, E., Sack, M., Nicholson, L., Fischer, R., Christou, P. (2005) Recent progress in plantibody technology. Curr. Pharm. Design 11, 2439-2457
Van Droogenbroeck, B., Cao, J., Stadlmann, J., Altmann, F., Colanesi, S., Hilmer, S., Robinson, D. G., Van Lerberge, E., Terryn, N., Van Montagu, M., Liang, M., Depicker, A., De Jaeger, G. (2007) High level accumulation of functional scFv Fc antibodies in Arabidopsis seeds: aberrant localization and underglycosylation of the recombinant proteins. Proc. Natl. Acad. Sci. 104, 1430-1435
